Complete Analysis of New Psychoactive Substances Using NMR
Introduction
Each year, more new psychoactive substances (NPS) are being formulated and produced under non-regulated conditions. These substances can be purchased legally by consumers, which pose an unpredictable risk to health and a challenge to law enforcement and medical personnel. There can be a high degree of structural similarity between illegal entities and legal analogues, regio-isomers for example, which means that the analytical data used during drug cases should be able to discern between those compounds with certainty.
This article shows how critical nuclear magnetic resonance (NMR) is for the unequivocal structural identification of illegal drugs and legal highs. The article demonstrates how DOSY experiments can be used along with computer-aided structure elucidation of the various compounds that make up synthetic drugs such as China White. It also shows how the structural information obtained can be used to streamline the identification and quantification of compounds present in recreational drug substances through the use of library search and matching tools. Figure 1 shows the NPS identified for the first time in the present year and the NPS identified in previous years.
Results and Discussion
Ketamine – Confirming Regioisomerism
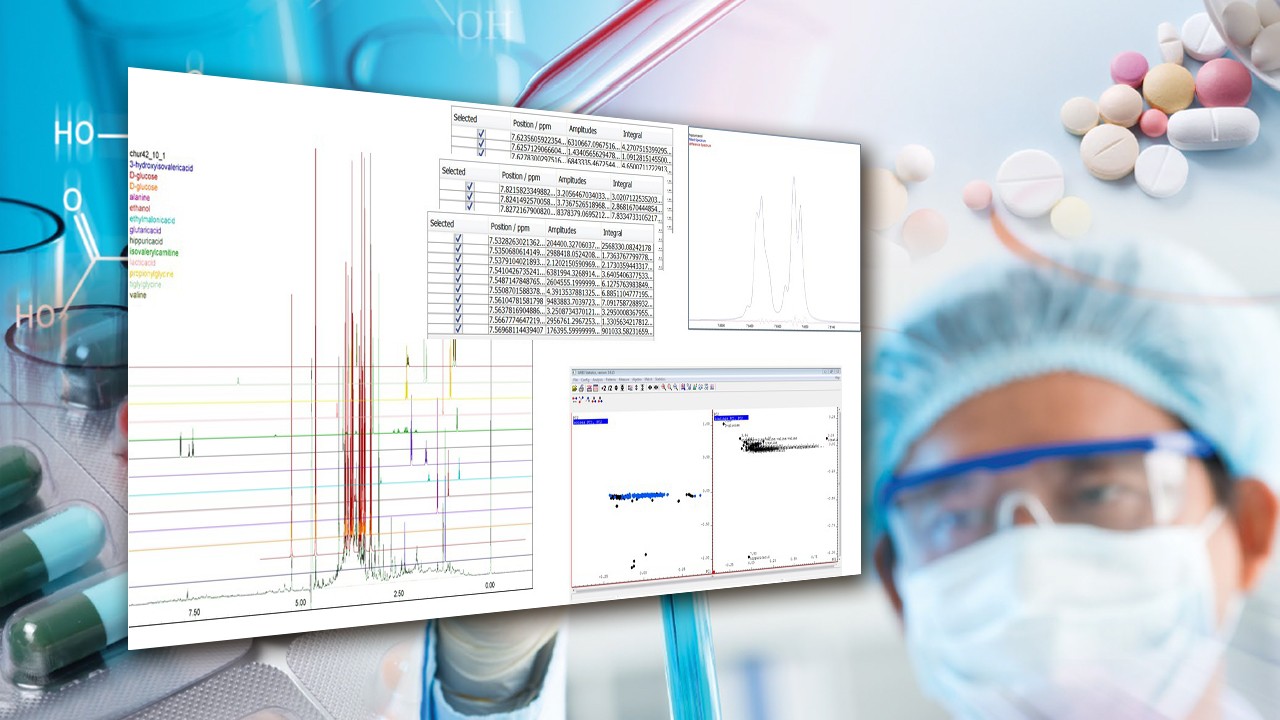
In order to verify the structure (class B compound) and exclude regioisomers (non-classified), NMR was used to analyze a ketamine sample. Verification was achieved using CMC-se software, including long-range HC correlations. The use of the new structure drawing tool for verification is shown in Figure 2.
Figure 3 shows the separation of compounds (A and B) with DOSY, and Figures 3a to 3c show the elucidation of A using CMC-se. The same methodology was carried out for compound B, which was found to be benzocaine.
Both the structure and the NMR spectra for benzocaine and methiopropamine were entered into a database. A method was developed for the identification and quantification of these compounds in mixtures using Assure Raw Material Screening (Assure RMS), which can be run in complete automation, from acquisition through to report creation.
By applying the newly developed Assure RMS method, further examination of other batches of China White can be carried out just with the push of a button, thereby significantly reducing the time taken for analysis.
Conclusion
NMR is an critical tool for complete analysis of NPS. NPS analysis using CMC-se software offers a number of benefits such as elucidation of NPS from scratch, differentiation of regioisomers and structure verification. It also eliminates bias and tunnel effect and allows exploration of the entire chemical space. The NPS mixture analysis workflow involved DOSY for compound separation; CMC-se of one compound at a time; and Assure RMS for compound identification, databasing and quantification.
References
[1] ‘Global Synthetic Drugs Assessment’ report, UNODC,
http://www.unodc.org/documents/southeastasiaandpacific/2014/05/gsda/2014_Global_Synthetic_Drugs_Assessment_embargoed_Tokyo_web.pdf, accessed 18Dec14
[2] http://www.bruker.com/products/mr/nmr/nmr-software/software/complete-molecular-confidence/cmc-se/overview.html, accessed 18Dec14
[3] http://www.bruker.com/products/mr/nmr/nmr-software/software/assure/overview.html, accessed 18Dec14
Acknowledgement
Produced from materials authored by Anna Codina1, Peter Gierth1, Chris Stapleton1, Andrew Kirkham1, Julia Smith1, Nicola Tonge1, Nora Kouris1, Brian Marquez2, Andrew Gibbs1 and John Ramsey3.
- Bruker UK Limited, Banner Lane, Coventry, CV4 9GH, UK
- Bruker BioSpin, 15 Fortune Drive, Billerica MA 01821, USA
- TICTAC Communications Ltd.