
Higher Order Structure
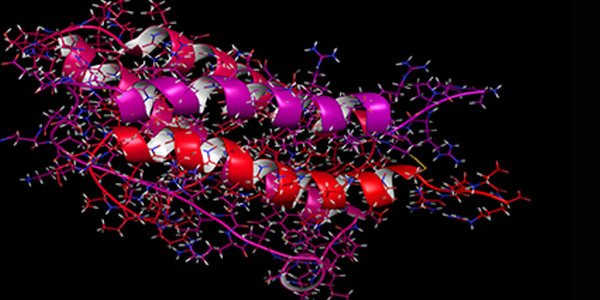
Unlike small-molecule drugs, biologics are produced in living systems, processing not only primary, but secondary, tertiary and possibly quaternary structures. Determination of a biologic drug's higher order structure (HOS) is critical to ensure efficacy, stability, and safety.
NMR is perfectly suited to evaluate protein structures and assess whether critical changes have occurred in a biotherapeutic drug during development and manufacturing. Due to its intrinsically high information content, NMR is proving to be a uniquely valuable tool for the evaluation of HOS, thus reducing the number of techniques needed to characterize biotherapeutic drugs. Recent advances in data acquisition and analysis mean that it is now possible to study intact materials (including monoclonal antibodies) at natural abundance, under conditions that are physiologically relevant. The new MbioHOS software offers a tool box of 1D and 2D method to evaluate the higher order structure of biotherapeutic drugs. Additionally, the assessment of similarity between a biosimilar drug and its reference material is now fast and easy, allowing rapid pass / fail evaluations to be made.
Using FT-IR as an Orthogonal Technique in HOS
FT-IR strenghtens the portfolio of orthogonal techniques for HOS analysis by giving insight into the secondary structure of proteins. The analysis is performed rapidly (~1 min/sample) and is characterized by a high tolerance against varying sample properties such as protein concentration, buffer components and excipients. These attributes make the method an optimal tool for monitoring the conformation of biopharmaceutical proteins during the development phase and for routine quality control.
This example application shows the IR spectra of an antibody at different temperatures (25-95° C). As indicated by the unchanged amide I band shape the protein maintains its native-sheet structure up to ~60° C. Further heating causes a decrease in intensity, which indicates the unfolding of intramolecular-β-sheets.
LabScape
Service & Life Cycle Support for Magnetic Resonance and Preclinical Imaging
Bruker’s commitment to provide customers with unparalleled help throughout the buying cycle, from initial inquiry to evaluation, installation, and the lifetime of the instrument is now characterized by the LabScape service concept.
LabScape Maintenance Agreements, On-Site On-Demand and Enhance Your Lab are designed to offer a new approach to maintenance and service for the modern laboratory