Intro
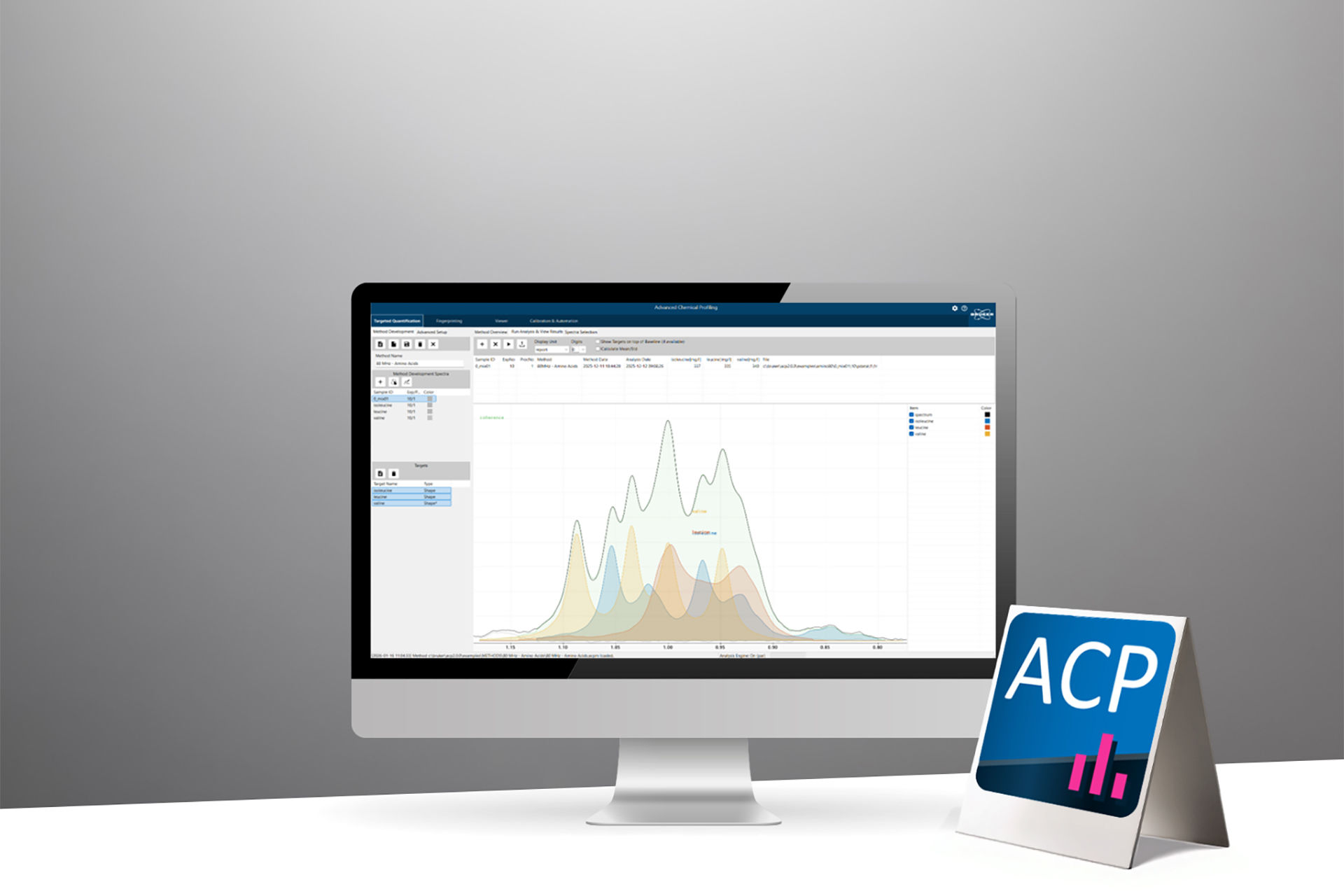
ACP 2.0 software empowers laboratories to implement push‑button, end‑to‑end automated NMR workflows across the entire product lifecycle, from early research through development and manufacturing. It redefines how analytical and testing facilities can leverage NMR technology by enabling harmonized workflows, significantly improving operational efficiency, and reducing costs through standardized, GMP‑ready, operator‑independent analysis, seamlessly connecting sample to final reporting.
Webinar Overview
ACP 2.0 enables fully automated end‑to‑end qNMR workflows, from acquisition, processing, identification, quantification, reporting, to result submission. With advanced signal modelling and fitting, high‑performance baseline handling, pass/fail evaluation, a custom calculation tool, and customizable reports, ACP 2.0 supports analytical challenges in industrial, biopharma and academia environments. It facilitates methods harmonization and transfer, allowing experts to develop robust workflows and deploy them to non‑expert environments. This webinar highlights core features, workflows, and use cases demonstrating how ACP 2.0 simplifies analysis and improves efficiency in quantitative NMR regardless of the sample’s complexity.
Wednesday, May 20 2026
8 AM PDT, 11 AM EDT, 5 PM CEST
What to Expect
In this webinar you will learn about the features and benefits of ACP2.0 software and how the new high-performance fitting capabilities enable the automation of any quantitative NMR workflow from simple to highly complex in the chemical & biopharma industry as well as in academic environments focusing on metabolomic research.
Key Learning Points
Automated End to End Workflow
- Delivers fully automated NMR analysis from sample to report, ensuring consistent, expert level results across any lab.
Standardized Decision Making
- Embeds robust pass/fail logic into workflows, eliminating subjective interpretation and ensuring harmonized global decision making.
High Throughput, Compliant Analysis
- Accelerates complex workflows with on the fly or batch processing, boosting productivity and reducing time to decision while supporting regulatory compliance.
Precision Through High Performance Algorithms
- Applies advanced fitting and deconvolution methods to deliver accurate, reliable quantification across even the most complex spectra.
Who Should Attend?
- R&D Professionals
- Analytical Scientists
- Lab Managers
- Principal Investigators
- QC Managers