Quality Control
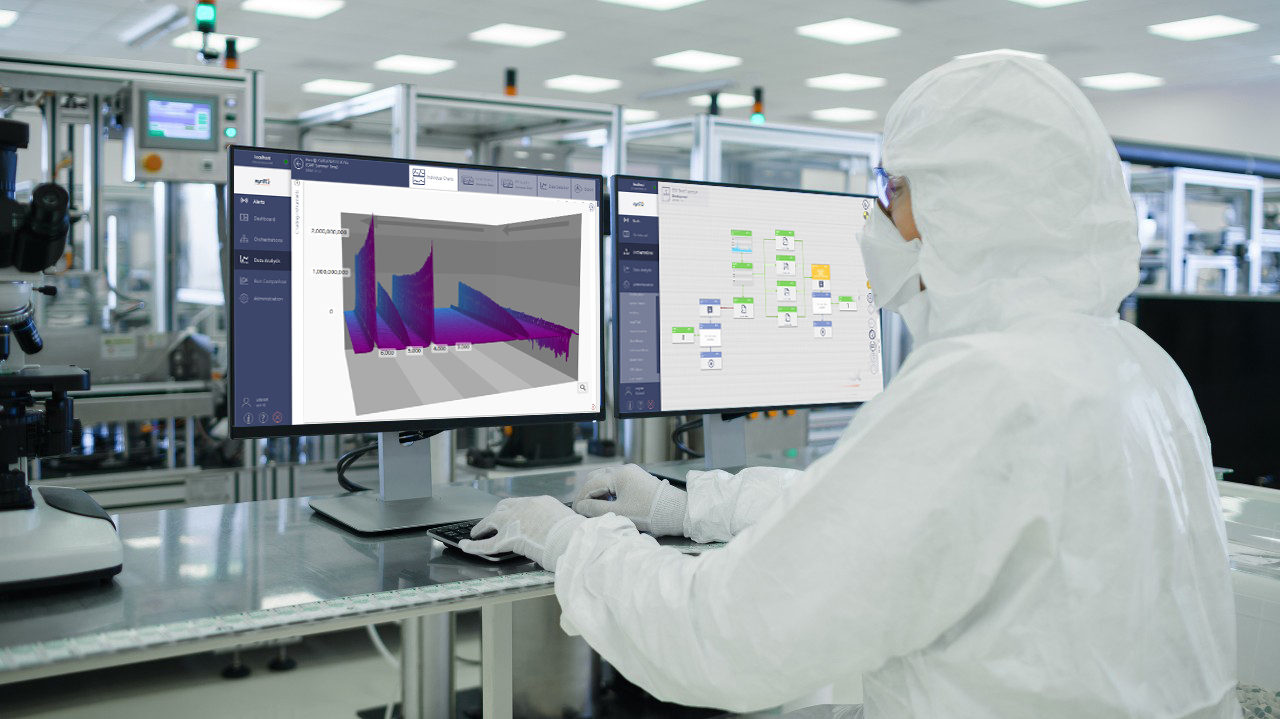
Quality control (QC) scientists are under scrutiny as they work to reduce risk in production, achieve rapid product release, and eliminate defects, impurities, and contamination, from raw materials through to finished product - as well as supporting relevant quality assurance (QA) and regulatory standards.
With increasingly stringent regulations applying to the pharma and biopharma industry, modern analytical technologies are playing a more and more important role, supporting QC scientists as they implement and improve control strategies to ensure that the highest quality standards are reached and maintained. Against this background, instruments that are designed for easy implementation of standard operating procedures (SOPs) in a GMP/GLP/cGMP environment and, where appropriate, the inclusion of installation and operation qualification (IQ/OQ) documentation and 21 CFR part 11 compliant software plays an important role.
Bruker is providing a comprehensive range of analytical systems, helping pharma QC scientists to manage their workload by providing tools for QC/QA applications that include: microanalysis by FT-IR/Raman microscopy; microbial identification by MALDI-TOF-MS; determining compound identity and quantitation by NMR; raw material identification by XRD and XRF; counterfeit analysis by FT-IR; analysis of APIs, additives and impurities by LC-MS, NMR, and XRF; manufacturing process control (PAT) by NMR and XRF.