Modern Pharmaceutical Quality Control
Highlights
End‑to‑end NMR Solutions for Compliant Quality Control
Modern pharmaceutical QC increasingly relies on analytical platforms that deliver reproducible, information‑rich results with minimal operational burden. Within this framework, Bruker’s NMR solutions provide a unified environment combining robust hardware, compliance‑ready infrastructure, and advanced automation capabilities.
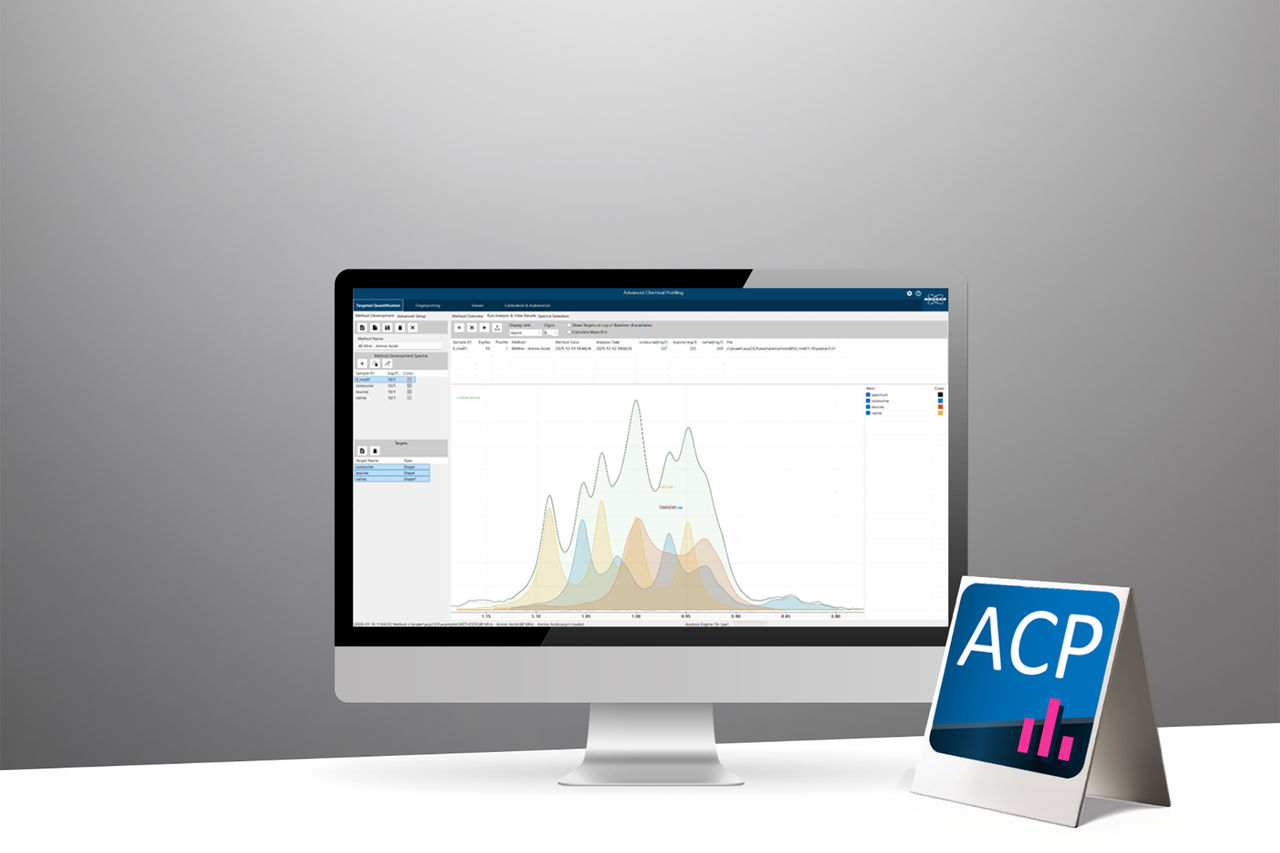
A central component of this ecosystem is the new Advanced Chemical Profiling (ACP) software, which integrates seamlessly into routine QC workflows without becoming the narrative centerpiece. ACP provides fully automated acquisition‑to‑report sequences, supporting both qualitative identity testing and high‑precision quantitative assays. Methods are implemented in a few structured steps within an integrated interface, enabling laboratories to reduce manual data handling while preserving the flexibility required for diverse regulatory environments. When combined with the Bruker GxP readiness kits, these workflows can be deployed directly in GMP/GLP settings, ensuring traceability, auditability, and consistent execution by non‑expert staff.
Unlocking the Robustness of NMR for Modern QC
At the analytical core, NMR spectroscopy remains the primary driver of robustness across the QC lifecycle. From raw material testing to the release of high-value drug products, NMR can be tuned for both small and large molecules, providing data-rich information that enables a wealth of applications. It delivers reportable values with confidence from simple but ultra specific qualitative procedures to complex, multi-attribute quantitative testing with unmatched robustness.
As a primary, absolute measurement technique, NMR aligns naturally with the modern framework of Analytical Quality by Design (AQbD). By leveraging technology‑inherent justifications and platform procedures, NMR reduces the need for extensive risk assessments, shortens method development, and simplifies procedure validation and transfer, in direct alignment with the recent concepts introduced in ICH Q14 and ICH Q2(R2). Its contactless nature eliminates stringent requirements for system conditioning, cleaning, and identical reference standards, thereby reducing time‑to‑result while improving operational reproducibility.
NMR also contributes to sustainability and operational efficiency. The absence of solvent consumption significantly reduces laboratory waste, while the stable, calibration‑free measurement principle supports predictable long‑term performance in routine testing environments.
Support
LabScape
Service & Life Cycle Support for Magnetic Resonance and Preclinical Imaging
Bruker’s commitment to provide customers with unparalleled help throughout the buying cycle, from initial inquiry to evaluation, installation, and the lifetime of the instrument is now characterized by the LabScape service concept.
LabScape Maintenance Agreements, On-Site On-Demand and Enhance Your Lab are designed to offer a new approach to maintenance and service for the modern laboratory