
PhenoRisk PACS™ RuO*
Multi-organ phenomic risk screening for research in Post-acute COVID Syndrome
PhenoRisk for research in Post-acute COVID Syndrome RuO
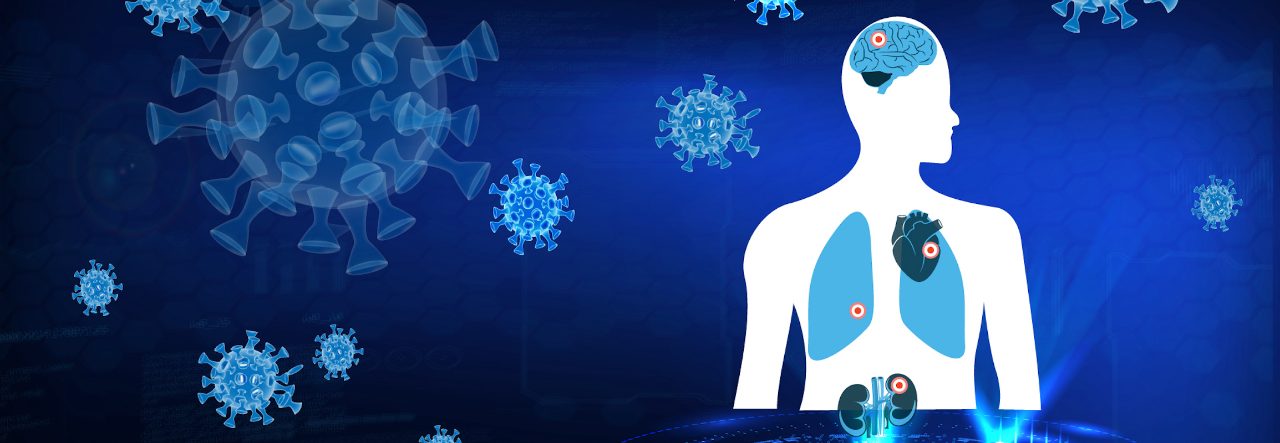
New plasma/serum quantification package detects and quantifies 16 different metabolites and ratios thereof in the context of research in COVID and the Post-Acute COVID-19 Syndrome (PACS). It indicates the potential for providing personalized risk information related to cardiovascular disorders, Type II Diabetes, kidney disorders and the overall inflammatory status.
PhenoRisk PACS™ enables highly reproducible quantitative multiplexed testing supporting researchers in the early detection of clinically well-characterized risk markers.
PhenoRisk PACS™ is a RuO solution for Bruker’s standardized and automated Avance IVDr NMR platform enabling researchers in-depth characterization of pathomechanisms of SARS-CoV-2, even in asymptomatic individuals, thus having the potential the potential to acting as a multi-organ risk screening for organ dysfunction after SARS-CoV-2 infection in a single laboratory test.
PhenoRisk PACS™ might enables the study of SARS-CoV-2 triggered metabolic phenoconversion, defined as transient or persistent systemic change of the molecular signature in human blood after acute SARS-CoV-2 infection. Subsequent phenoreversion, indicated by normalization of the metabolic signature, can be detected by PhenoRisk PACS™ and may mark PACS recovery.
PhenoRisk PACS™ provides insight into unique (RuO) biomarkers, exclusively assessable by NMR technology (Glyc A/B, SPC) for deeper information on ongoing inflammation processes.
PhenoRisk PACS™ deepens the understanding of the multisystemic nature of SARS-CoV-2 and PACS thus supporting basic and translational clinical research as well as the development of impactful SARS-CoV-2 treatments by:
- Carrying it out on Bruker’s metabolic profiling Avance IVDr NMR platform, it provides easy to operate and non-destructive, label-free multiplexed testing with minimal hands-on time.
- Analyzing in a single EDTA plasma or serum human sample, in solely one run, under standardized and automated conditions at affordable costs.
- Seamlessly complementing Bruker’s RuO biomarker panel portfolio, providing in-depth understanding of lipoprotein profiles (B.I.LISA, 112 analytes) and for quantification of small metabolites (B.I.QuantPS2.0, 40 metabolites).
*Bruker NMR Instruments are for Research Use Only. Not for Use in Clinical Diagnostic Procedures
Features
Includes
- Concentration of 16 metabolites and ratios thereof (free metabolites, no protein denaturing done) with indication of the 95% concentration ranges that were derived from the respective healthy model cohorts.
- In the analysis report, the metabolites are grouped according to the indications diabetes, kidney disorders, cardio-vascular disease (CVD) and inflammation.
- Information about the quality of the data in terms of NMR instrumentation, experiments, spectral quality, sample preparation, matrix identity test and contamination check (For more detailed information please see the Bruker IVDr BioBank QC).
- The report contains comprehensive explanation pages and a literature bibliography.
- In addition to pdf reports and xml result files, results are also directly saved in csv format allowing direct input for follow-up calculations.
Benefits
- Ease of use, simple and rapid sample preparation
- Fully automated quantification of 16 metabolites and ratios thereof
- Different analyte classes can be quantified simultaneously in a single run
- Absolute concentration for each metabolite is given due to the calibration with one Quantification Reference Sample (provided with B.I.Methods2.5™)
- Rapid analysis: >70 samples can be prepared, measured, and analyzed daily
- Retrospective analysis possible if the sample was prepared and measured using B.I.Methods2.5™
- Works on EDTA plasma and serum samples
Applications
PhenoRisk PACS™ has the potential to help researchers with the discrimination of SARS-CoV-2 / PACS patients from healthy or recovered individuals, which could be important amongst others for translational research on new treatment approaches. This NMR-based, molecular phenomics monitoring with PhenoRisk PACS™ supports research in revealing:
- disease progression in acute infection
- patient’s partial or full recovery
- severity of the disease
- treatment outcome
Specifications
- Bruker Avance IVDr NMR platform or compatible AVANCE™ III HD or AVANCE™ NEO 600 MHz systems
- Use of B.I.Methods2.5™ module and SOPs for plasma/serum for sample preparation and measurement
- Regular quality control of the absolute temperature, solvent suppression and quantification reference sample (preferably daily using automated quality control tool included in B.I.Methods2.5™)
- Access to Bruker Data Analysis server for fully automated remote analysis (transfer of spectra after measurement to Bruker server via private ftp, back-transfer of result PDF report and XML file)
Download Unlocking our clinical understanding of SARS-CoV-2 infection
LabScape
Service & Life Cycle Support for Magnetic Resonance and Preclinical Imaging
Bruker’s commitment to provide customers with unparalleled help throughout the buying cycle, from initial inquiry to evaluation, installation, and the lifetime of the instrument is now characterized by the LabScape service concept.
LabScape Maintenance Agreements, On-Site On-Demand and Enhance Your Lab are designed to offer a new approach to maintenance and service for the modern laboratory