MICRONAUT IVD System
MICRONAUT IVD system offers a range of antibiotic resistance and specialty susceptibility testing
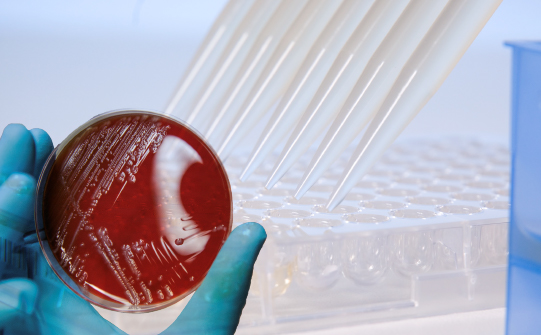
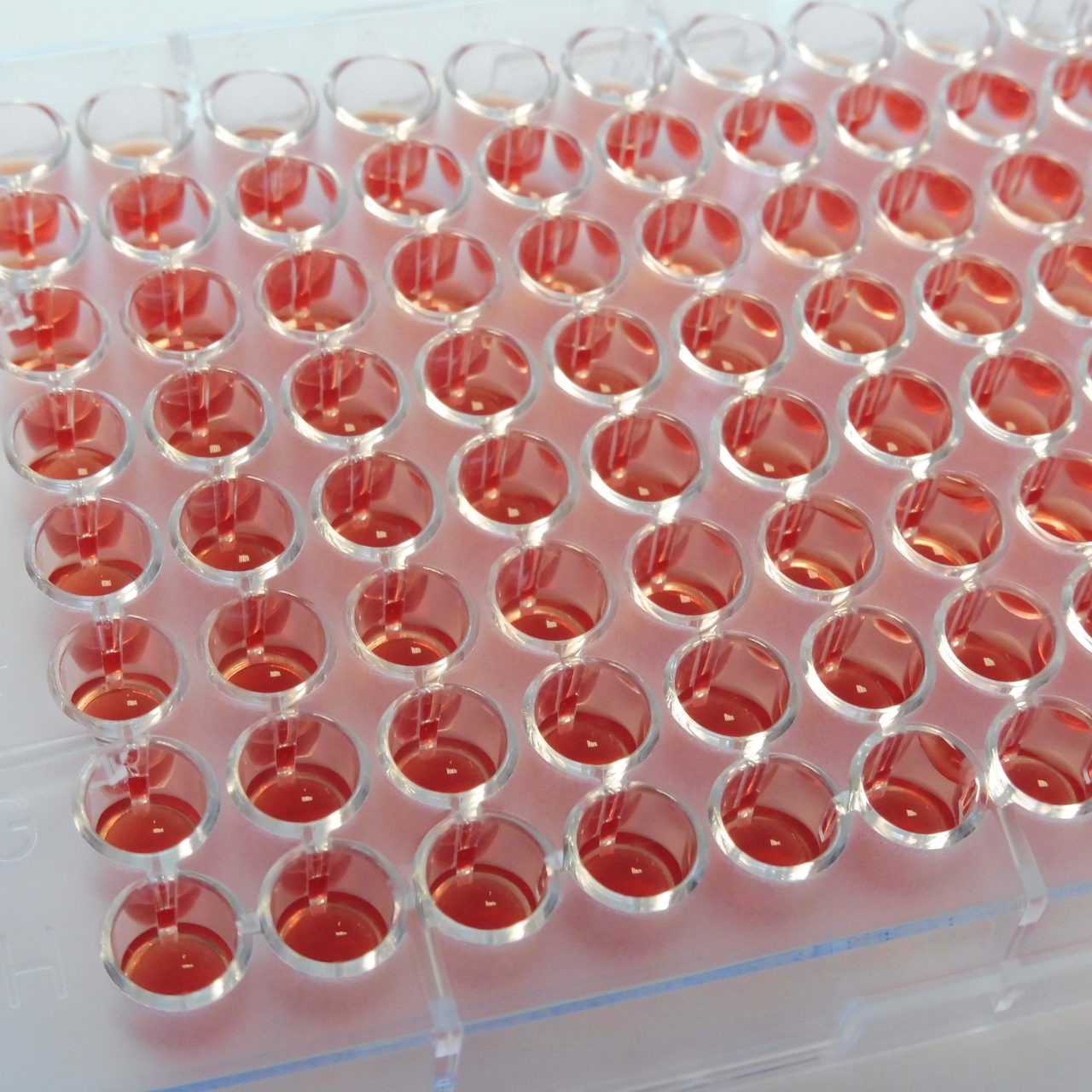
The MICRONAUT IVD System encompasses a range of plates for detection of resistance mechanisms and specific resistances of clinical and/or epidemiological importance, including antimicrobial susceptibility testing (AST) with standard minimum inhibitory concentration (MIC) plates, covering an extended spectrum of organisms in microbiology.
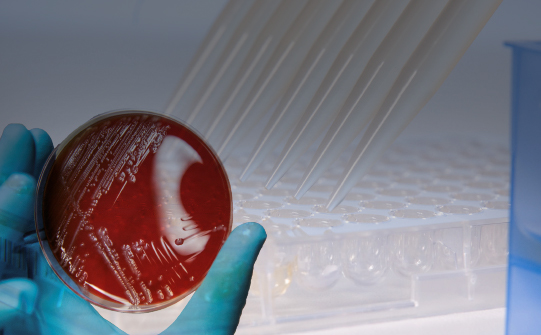
Routine microbiology laboratories are in need of an efficient tool for phenotypic detection of important antimicrobial resistance mechanisms, such as resistance genes encoding for certain ß-lactamases (e.g. ESBL or carbapenemases). These mechanisms are quickly and efficiently transferred between pathogens, causing concern over the effectiveness of key antibiotics.
Broad resistance coverage
The MICRONAUT system provides antimicrobial susceptibility testing (AST/AFST) either with standard MIC plates with a dedicated layout or with individual customized layouts.
In addition, the MICRONAUT system offers the detection of clinically relevant resistant mechanisms in gram-negative and gram-positive bacteria.
The flexibility of the MICRONAUT system allows laboratories to adjust their testing profiles to the rapid spread of newly emerging resistances.
MICRONAUT AST
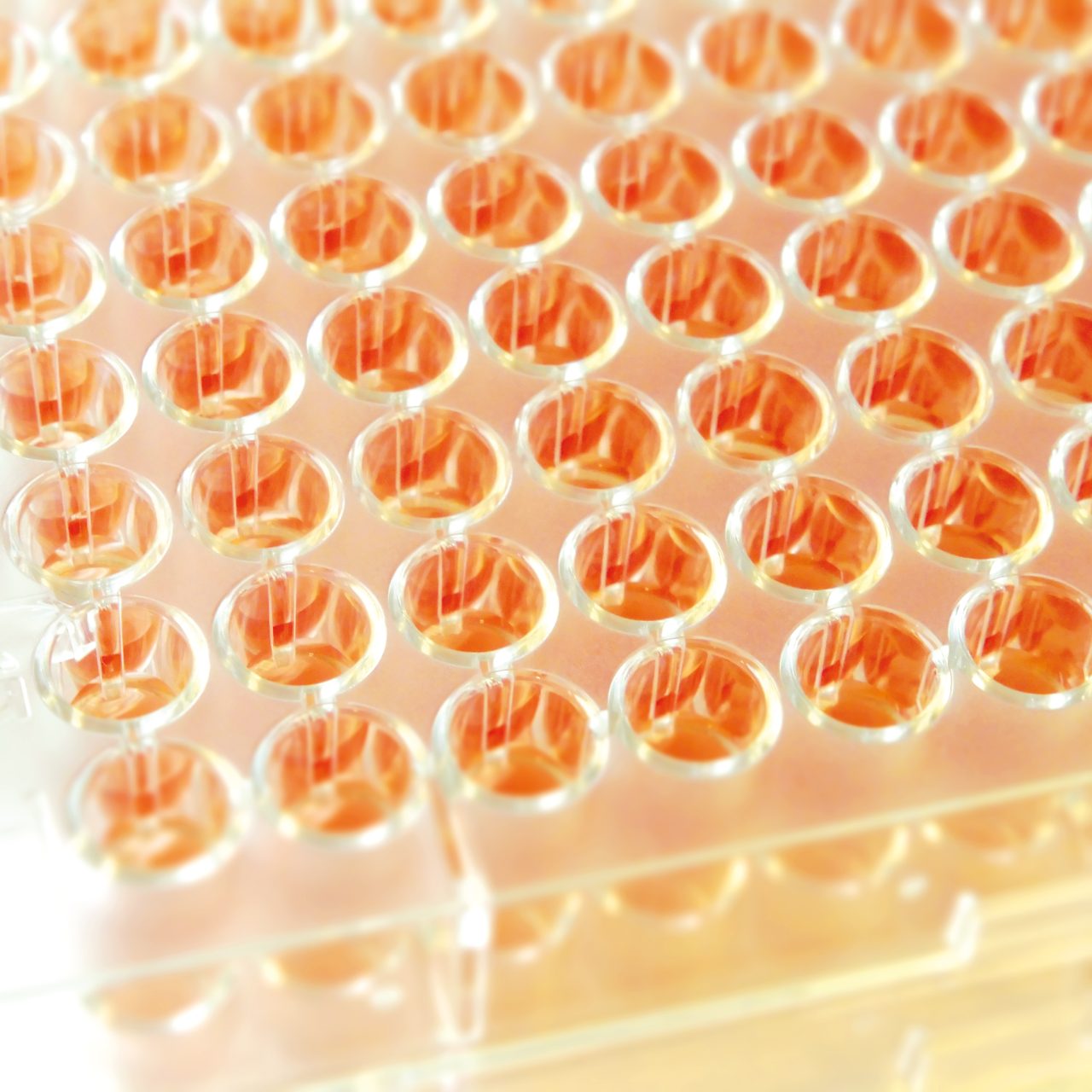
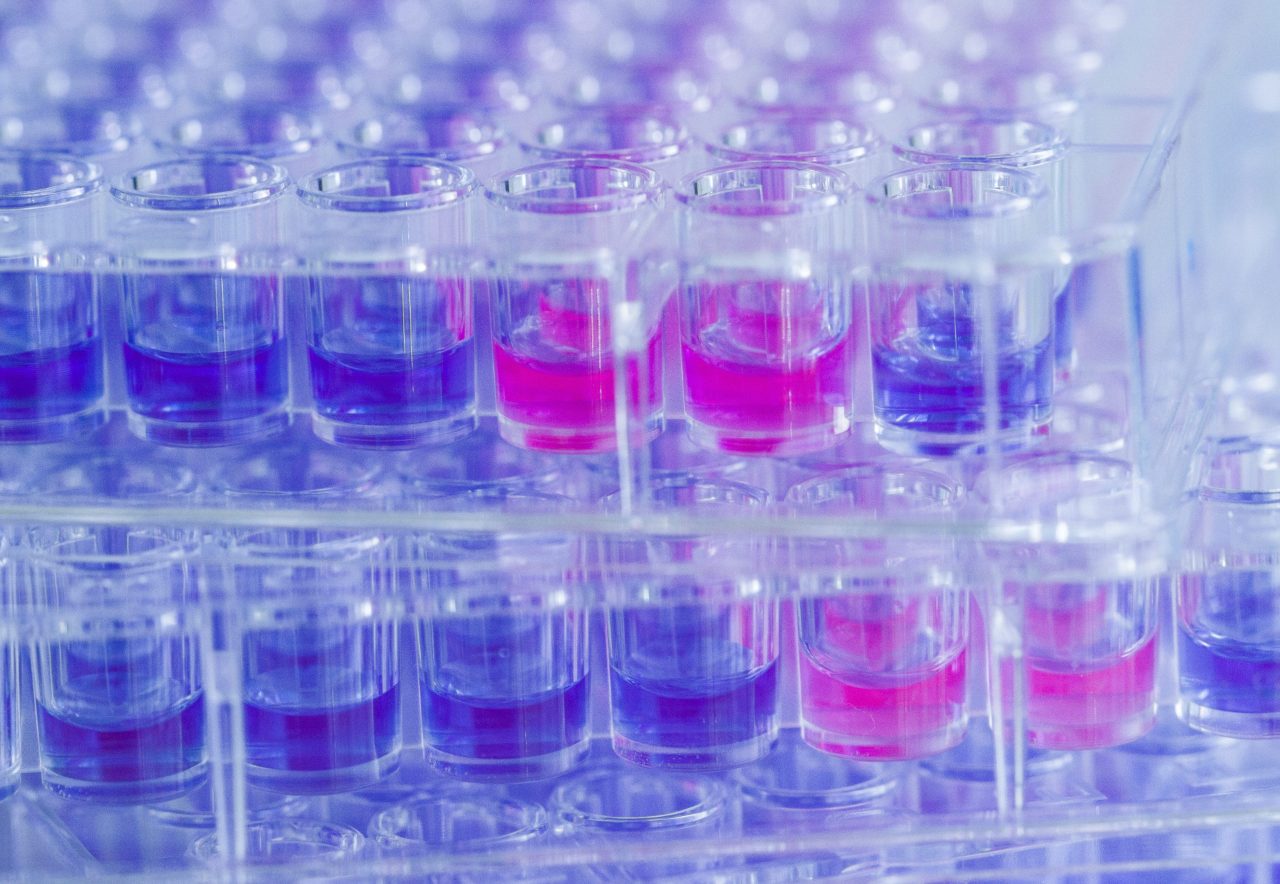
The test principle of the MICRONAUT system for AST is based on phenotypic detection of resistance as expressed by microbial growth in the presence of antibiotic compounds. The broth microdilution (BMD) procedure is a standardized method and a worldwide accepted reference system for determination of true MIC values.
Users can choose preferred standards, such as EUCAST or CLSI, and compose customized antibiograms from more than 200 antibiotics, or select from a range of standard layouts. MICRONAUT-S and MICRONAUT-AM plates, as well as MIC-Strip manual susceptibility testing, are designed for in vitro diagnostic use.
Please contact your local representative for availability in your country.
Not for sale in the USA.
As of January 2022, the Legal Manufacturer of the mentioned Mueller Hinton broth and all MICRONAUT products is Bruker Daltonics GmbH & Co. KG.