Application Note: Processive 8-nm Kinesin Stepping Resolved with the NanoTracker
The combination of optical tweezers with back-focal-plane detection allows for the simultaneous position and force detection at nm and pN resolution, respectively. The NanoTracker is the first off-the-shelf optical tweezers system to deliver a complete solution providing the capacity of sub-picoNewton force measurements together with high spatial and temporal resolution of the position of the molecule. The instrument furthermore merges versatile methodologies by combining ultra-stable dual trap optical tweezers with DIC imaging and fluorescence visualization.
In this application note, it is demonstrated that the tracking of the processive movement of motor proteins such as kinesin can be easily performed, yielding information on kinetics, step sizes, and forces associated with the movement. With the NanoTracker, Bruker provides another tool to perform ultra-sensitive measurements on the single molecule level for life scientists.
Readers can expect to learn about:
- The configuration and calibration of the NanoTracker Optical Tweezers system for performing motor-protein experiments, showcasing the easy operation and the seamless integration with optical microscopy;
- Experimental assay and detailled discussion of the results showing the outstanding resolution of the system; and
- The exceptional potential of the NanoTracker for single-molecule experiments under native conditions.
KEYWORDS: Optical Tweezers; NanoTracker; Fluorescence Microscopy; Optical Integration; Force Measurements; Motor Proteins; Kinesin; Microtubules; Nanomanipulation; Life Science
Introduction
Since the mid 1990s, a new era of biomechanical studies has been launched by the development of optical methods. With its high force sensitivity, optical tweezers promised to be a potent tool for the investigation of single molecules. The introduction of optical probes such as polystyrene and silica beads allowed biological macromolecules to be investigated with optical tweezers. The molecules of interest were bound to the bead and allowed to interact with other beads or a functionalised cover slip surface. In addition, optical tweezers were used as a single-molecule technique to investigate the mechanical function of proteins involved in intracellular transport and other processive activities, such as DNA and RNA replication. The most intensive applications so far of optical tweezers have focused on linear motors which move along a linear track. This work resulted in a large number of reports published on kinesin mechanics. With the development of experimental setups for the detection of single 8 nm steps of kinesin on microtubule filaments 1, the way was paved for the extensive characterisation of the kinetic and biochemical function of this motor protein. The stalling force 1, 2, the rate of the movement depending on the ATP concentration 2, 3 and the impact of load on the catalysis of ATP 3, 4, to name a few, have all been characterised by optical tweezers. In this report, we demonstrate the kinesin movement on microtubules with the NanoTracker™ demonstrating its high spatial resolution. The NanoTracker™ simultaneously records displacement and force data, yielding steps and stalling forces in a single measurement.
Kinesin: a motor protein walking processively on microtubules
The cell is a highly organised compartment where nutrients, signal messengers and building blocks need to be delivered to the location of interest. While small molecules traverse the cell purely with the help of brownian motion, macromolecules and vesicles need to be transported via motor proteins. The most prominent motor proteins are kinesin, myosin and dynein, of which the two latter ones are also responsible for cell motility. Kinesins are a group of related proteins that walk stepwise along microtubules. They are responsible for the transport of chromosomes during mitosis as well as organelle and vesicle transport in the cytoplasm of the cell. The well-studied conventional kinesin-l is a homodimer that carries organelles towards the periphery of the cell. The transport is driven by ATP hydrolysis, which causes the kinesin to walk processively along the microtubules in steps of 8 nm 1, the size of a tubulin dimer (see figure 1) 5. A single kinesin molecule can take several hundred steps before detaching, even against opposing loads as high as —7 PN 3.
In order to measure single kinesin steps, an assay was chosen in which microtubule filaments are attached to the glass surface of the flow cell via antibodies 6. To study the interaction, single kinesins are bound to caseincoated microspheres, which are brought to the vicinity of the filaments by means of an optical trap. The processive stepping is then recorded on a NanoTracker™ setup at room temperature and at non-saturating ATP conditions.
Experimental Procedures
NanoTracker™ optical tweezers platform
For these experiments, the NanoTracker™ (see figure 2), a state-of-the-art optical tweezers platform including a sensitive detection system, was built onto a Zeiss AxioObserver.A1 equipped with epi-fluorescence. The NanoTracker™ allows for nanomanipulation of the trap in 3-D, crucial for the precise positioning of the beadcoupled kinesin onto the only 25 nm thick microtubule. Force and displacement measurements are performed using back-focal-plane interferometry of the scattered light from the bead on quadrant photodiodes. All trap and sample positioning, trap stiffness adjustment, detection calibration and data recording are conveniently controlled via the NanoTracker™ software.
The one-click calibration of the detection makes use of the power spectrum of the particle's movement inside the trap. A fit to the spectrum delivers data on the sensitivity of the detection (in nm/V) and the stiffness of the trap (in pN/nm). In a range of a few hundred nanometers measured from the center of the trap, there is a linear relationship between the particle's position and the restoring force acting on it (see figure 3).
Experimental assay
The microtubule filaments are attached to the bottom of the sample chamber via antibodies. The adsorption is verified with the NanoTracker™ using the integrated fluorescence microscope. Polystyrene beads (1.53 urn diameter) labelled with single kinesins are added to the flow cell and used for calibration of the trap. The laser intensity is adjusted to achieve a trap stiffness of around 0.05 pN/nm. At this stiffness and with the assumption that kinesin stepping stalls around 7 pN, the motor protein can progress in the trap for 140 nm, which is well within the linear detection range of the trap. All experiments are performed in BRB80 buffer (80 Pipes, 1 rnM MgC12, 1 rnM EGTA, pH 6.9), 10 PM ATP, 10 PM taxol and 0.16 mg/ml casein. For bright and stable fluorescence imaging, a glucose oxidase/peroxidase oxygen scavenging system is accessorily added to the solution.
In order to initiate the binding and the stepping of the kinesin on the microtubules, a trapped bead is approached towards a microtubule. The contact with the surface is observed as a displacement of the bead in the z-direction of the trap. The active bead usually binds to the microtubule within a few seconds and starts walking on the filament. The stepping is recorded at a sampling rate of 4 kHz.
Experimental Results
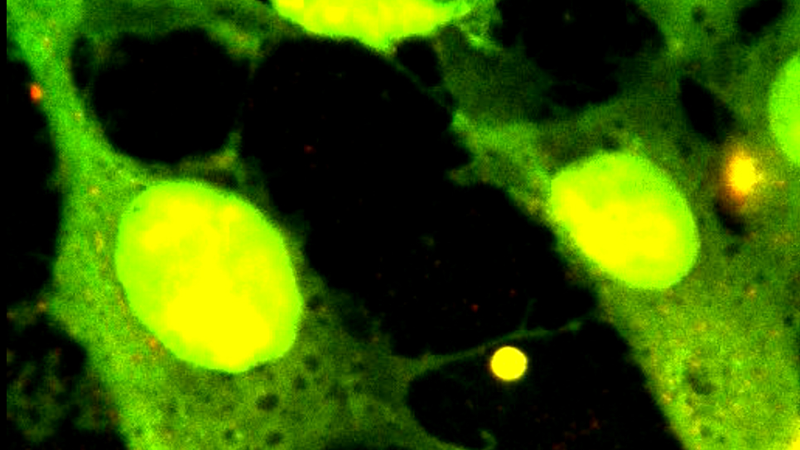
The proper binding of the microtubules (MT) to the coverslide surface is verified with epi-fluorescence. An image of single microtubules and bundles of them (higher brightness) is shown in figure 5.
When bringing a kinesin-coated bead towards a microtubule, the bead binds to the filament and progressively walks away from the center of the trap until the stalling force of the kinesin is reached.
For recording of single steps, the kinesin concentration was diluted such that at a chance of at least 90%, one kinesin is bound per bead at most. In practice, this will mean that only about 10% of the beads will be active. In figure 6B, a representitive trace of a single kinesin stepping on a microtubule filament is displayed. At low counteracting forces, the kinesin steps at a such high rate that single steps can hardly be resolved (see figure 6A). At larger displacements from the center of the trap (here 60 nm) and consequently larger counteracting force, the speed is noticably reduced and the steps can thus be clearly resolved. The four steps displayed here have a dwell time of around 0.1-1 s. This stepping speed was achieved by working at non-saturating ATP conditions (10 PM). The dependency of the stepping speed from the ATP concentration is described in detail in ref. 7
A trace of kinesin-labeled beads repeatedly binding to MT filaments and walking out of the trap until the stalling froce is reached is shown in figure 6A. As can be seen in this figure, the dwell periods between the steps lengthen with increased load: while the velocity of the stepping is constant at low forces, it steadily decreases with increasing load, until the bead finally tears off from the filament. In the experiments performed, the stalling force varied between 4 and 7 pN.
Conclusions
The combination of optical tweezers with back-focalplane detection allows for the simultaneous position and force detection at nm and PN resolution, respectively. The NanoTracker™ is the first off-the-shelf optical tweezers system to deliver a complete solution providing the capacity of sub-picoNewton force measurements together with high spatial and temporal resolution of the molecule's current position. The instrument furthermore merges versatile methodologies by combining ultra stable dual trap optical tweezers with DIC imaging and fluorescence visualisation.
Sophisticated single-molecule experiments can now consequently be carried out routinely by a wide range of life science researchers without the need to design and build custom instruments. In this report, it has been demonstrated that the tracking of the processive movement of motor proteins such as kinesin can be easily performed, yileding information on kinetics, step sizes and forces associated with the movement. With the NanoTracker™ , JPK Instruments provide another tool to perform ultra-sensitive measurements on the singlemolecule level for life scientists.
Acknowledgements
Many thanks to Volker Bormuth and Dr. Erik Schäffer from the Max-Planck-Institute of Molecular Cell Biology and Genetics and the Biotechnology Center in Dresden, respectively, for kindly providing the samples (kinesin-l, microtubules) and their continuing support regarding the experimental procedure.
Literature
- K Svoboda, CF Schmidt, BJ Schnapp and SM Block, Direct observation of kinesin stepping by optical trapping interferometry, Nature 365, 721 (1993).
- H Kojima, E Muto, H Higuchi, and T Yanagida, Mechanics of single kinesin molecules measured by optical trapping nanometry, Biophys J. 72, 2102 (1997).
- E Meyhöfer and J Howard, The force generated by a single kinesin molecule against an elastic load, Proc. Natl. Acad. Sci. U S. A. 92, 574 (1995).
- K Svoboda and SM Block, Force and velocity measured for single kinesin molecules, Cell 77, 773 (1994).
- S Ray, E Meyhöfer, RA Milligan and J Howard, Kinesin follows the microtubule's protofilament axis, J. Cell Biol. 121,1083 (1993).
- V Varga, J Helenius, K Tanaka, AA Hyman, TU Tanaka and J Howard, Yeast kinesin-8 depolymerizes microtubules in a length-dependent manner, Nature Cell Biol. 8, 957 (2006).
- SM Block, CL Asbury, JW Shaevitz and MJ Lang, Probing the kinesin reaction cycle with a 2D optical force clamp, Proc. Natl. Acad. Sci. U. S. A. 100, 2351 (2003).
LEARN MORE: