Positive Blood Culture Analysis
When every minute counts
Sepsis is a critical medical condition affecting over 30 million people worldwide each year. Timely identification of the responsible pathogen is crucial for improving patient outcomes, reducing morbidity and mortality, and positively impacting healthcare costs. Matrix-Assisted Laser Desorption/Ionization Time-Of-Flight Mass Spectrometry (MALDI-TOF MS) can accelerate the diagnostic process, cutting turnaround times by up to 48 hours when compared to conventional laboratory procedures.
traditional methods
therapy in sepsis patients
Actionable results based on one single sample run
Direct identification from Positive Blood Cultures
Bruker’s MBT Sepsityper® solution has been designed to work seamlessly with the MALDI Biotyper® (MBT). This powerful combination allows the identification of pathogens from positive blood cultures in just 15-20 minutes, presenting a cost-effective and efficient workflow that enhances patient care. The enhanced Sepsityper® Kit not only improves MBT identification rates but also helps reducing the time required to transition patients from empirical therapy to more targeted treatment approaches.
The MBT Sepsityper® solution is available for IVD-CE as well as US-IVD clinical workflows, and it is a very cost-effective alternative to molecular solutions.
Swift pathogen identification with the dedicated Sepsityper® Kit
Bruker’s Sepsityper® Kit delivers rapid and reliable species identification from positive blood cultures. The workflow requires less than 10 minutes of hands-on time, providing identification within 15-20 minutes after a positive blood culture alert. Moreover, multiple assays can be run simultaneously, streamlining laboratory processes. The results obtained through the MBT Sepsityper® solution empower physicians to effectively manage bloodstream infections, combat antimicrobial resistance, and enhance patient outcomes.
Dedicated Sepsityper® Kits are available for IVD-CE and US IVD clinical workflows
Fighting antimicrobial resistance
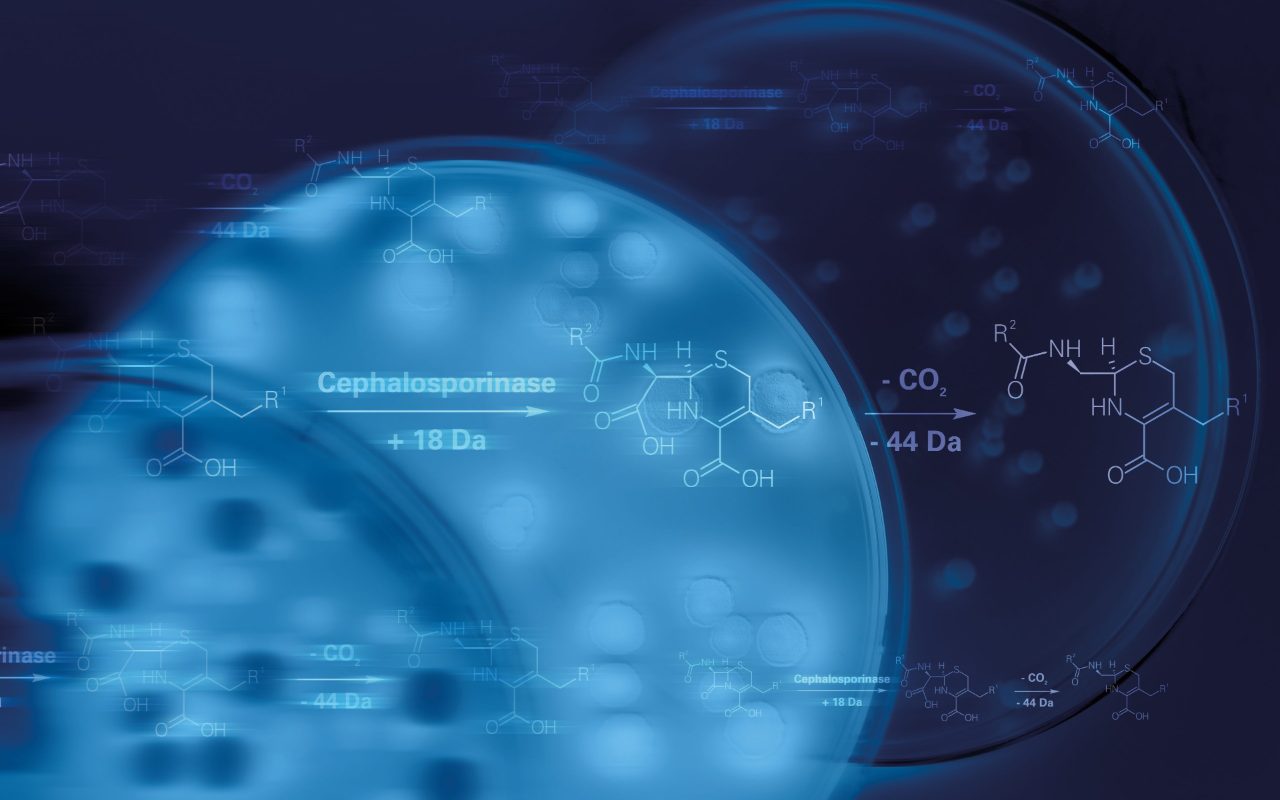
Detecting resistance to first-line antimicrobials is possible as part of the positive blood culture workflow. Bacterial cells isolated – using the Sepsityper® Kit - from flagged positive blood cultures can be employed in MBT STAR®-BL assays for beta-lactamase activity detection. Dedicated Kits facilitate rapid identification of bacteria and the detection of carbapenemase/cephalosporinase activity within a single workflow.
For clinical use, the MBT STAR®-BL assays are only available for IVD-CE workflows.
Sepsityper® solutions fitting to your clinical microbiology lab
Bruker’s Sepsityper® solution is available in two different configurations. Please contact your local representative for availability in your country:
MBT Sepsityper® IVD Kit and MBT HT Sepsityper IVD Module
Covering the entire IVD-CE reference library.
For professional use only. Not for sale in the USA.
-
MBT Sepsityper® IVD Kit (brochure)
(PDF, 1 MB)
-
Expert Insights (Reducing Time to Diagnosis Sepsis)
(PDF, 371 KB)
-
Expert Insights (Bologna Workflow - Improving patient outcomes with rapid, reliable microbial identification and resistance testing)
(PDF, 4 MB)
-
Whitepaper - Tackling the global sepsis crisis with advanced MALDI-TOF MS technology
(PDF, 301 KB)
-
Application Note - Comparison of different short culturing methods to the Rapid Sepsityper workflow: Microorganism identification from positive blood cultures
(PDF, 908 KB)
MBT Sepsityper® Kit US IVD and MALDI Biotyper® CA System software
Covering the entire FDA-cleared reference library.
Only for sale in the USA and Puerto Rico. For prescription use only.