Drug Discovery
Bringing a new drug into the market, from the first step to the final market introduction, is a time consuming, highly regulated, and expensive process, which can take a decade or more. Final success crucially depends on the early availability of accurate analytical results, fast enough for taking the right decisions at the beginning of the development, and minimizing late attrition rates.
Today’s drug development is mainly based on a rational approach where typically establishing the biological target to focus-on is the first key step. This target identification requires a deep understanding of the candidates´ properties to identify the most promising ones as quick and reliable as possible. The biological interplay and relevance of a protein and its ligand(s) require insights into both structural and affinity related aspects – all that is covered by Bruker’s broad portfolio in nuclear magnetic resonance (NMR), mass spectrometry (MS), single-crystal X-ray diffraction (SC-XRD), and surface plasmon resonance (SPR). In many cases, preclinical imaging delivers important upfront information if a potential target elicits a biological response that might deem to modulate the disease.
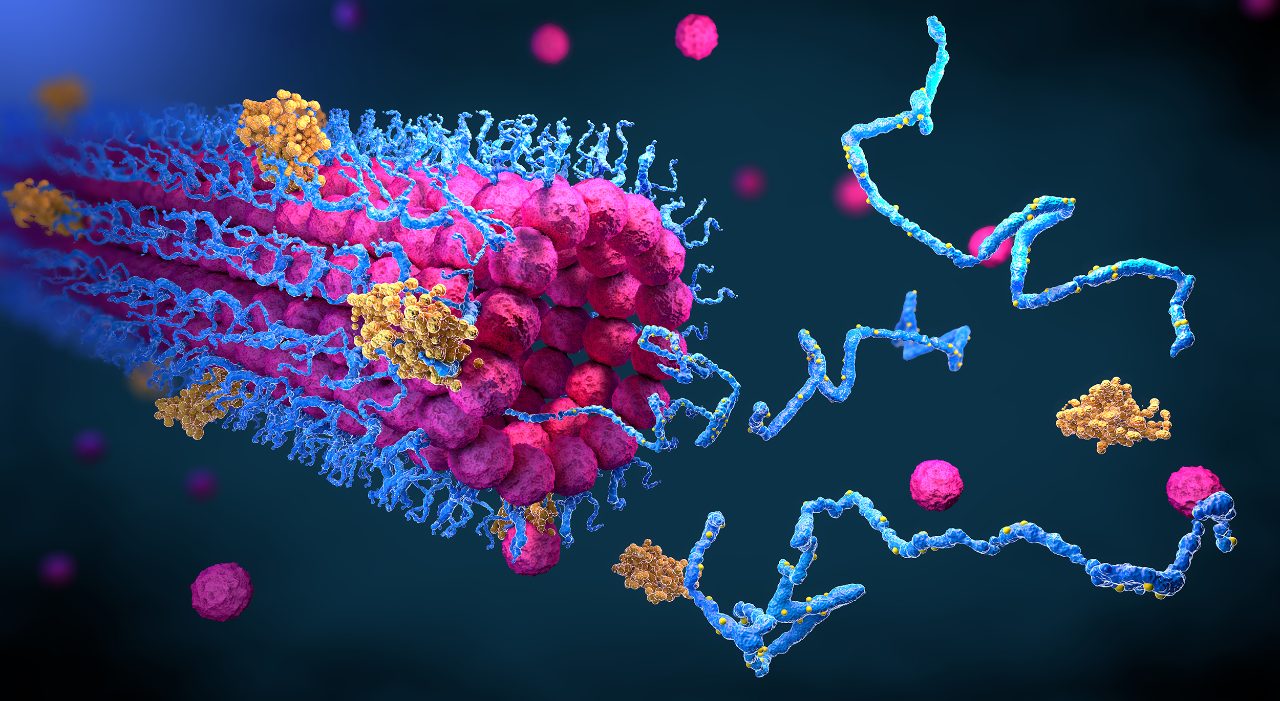
Once a biological target has been established, finding the most promising lead molecules is often seen as the next challenge. Typically, lead discovery is the identification of potential drug candidates – either small organic molecules or biologic assemblies with therapeutic potential.
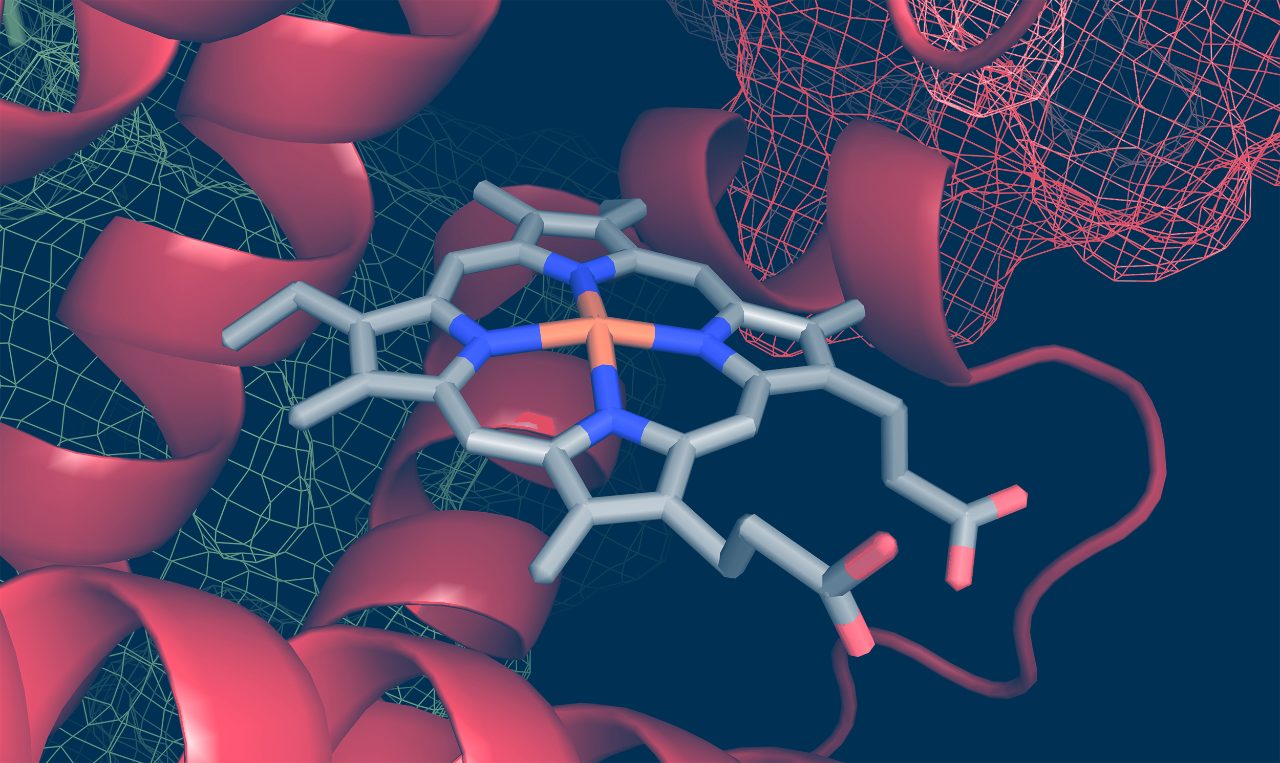
Rational drug design starts with the knowledge of the molecular structure of the target or reported active ligands. Both computational and empirical knowledge can be used to perform structure or ligand-based drug design (SBDD and LBDD, respectively). These approaches may be characterized by a comparably lower throughput requirement, but higher data density, e.g. about selectivity or the binding mode simultaneously. NMR, MS, SPR, Small-angle X-ray scattering (SAXS), and single-crystal X-ray diffraction (SC-XRD ) are techniques to mention here in the first place.
As the ongoing drug discovery process is time-sensitive, high throughput instrumentation with superior uptime, data quality, and assay flexibility is critical for success. Lead optimization requires advanced techniques allowing to investigate protein-ligand interactions from different perspectives at high data quality, yet with less throughput. The daily analytical support required for fast and successful medicinal chemistry is efficiently provided by our NMR, MS, SC-XRD solutions, and the combination of those. Whenever, throughout the entire drug discovery process, questions related to the details of the structure analysis are addressed, these methods deliver the high-quality answers, indispensable and in-time.
Today, efficient drug discovery includes detailed studies to understand ADME-Toxicity (absorption, distribution, metabolism, excretion, toxicity) at an early stage. We have developed dedicated toolboxes for our NMR, and MS solutions helping scientists to gain insight faster, avoid later phase failures, and comply with the most recent regulations.
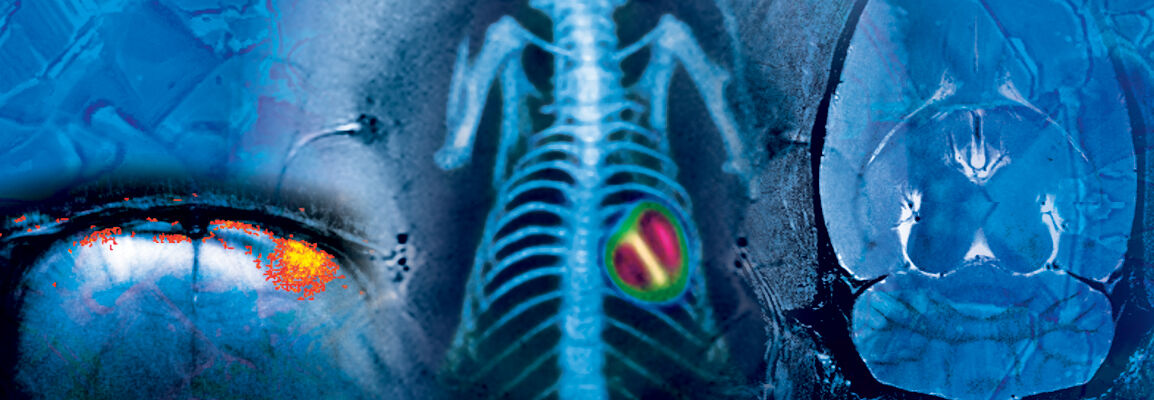
Accelerating decision-making and building confidence inefficacy of treatment can be achieved faster in the drug discovery process using modern preclinical imaging solutions. Our solutions for animal MRI and microCT are setting the industrial standards and help to maximize the insights and consequently to minimize the number of test animals required.