
Linking Synaptic Specificity to Hippocampal Circuit Function
Learn how molecular mechanisms of brain development shape the neuronal circuits that underlie behavior.
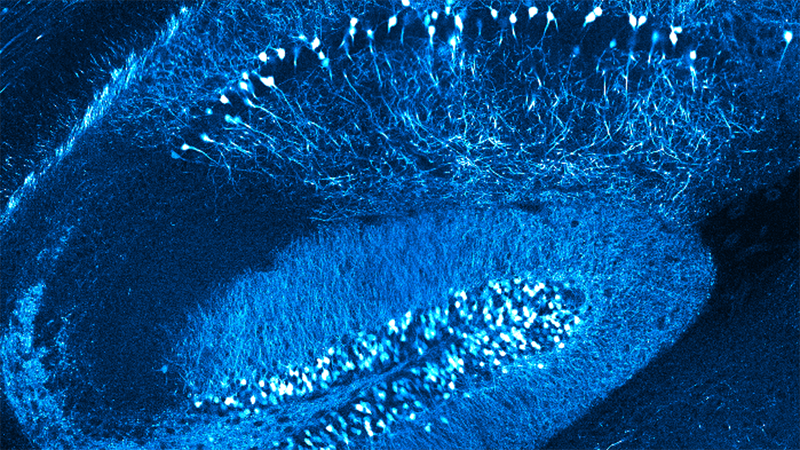
Learn from Dr. Heike Blockus about how multiphoton microscopy is utilized to explore the complexity of interactions occurring at synapses. Two-photon calcium imaging is used to better understand CA1 pyramid neurons in free-behaving mice with real-world applications.
Webinar Summary
The developmental transition between axon guidance and synapse formation is critical for circuit assembly but still poorly understood. We present evidence that this key transition can be regulated by axon guidance cues switching their function to regulate synaptogenesis with subcellular specificity.
While investigating specific molecular mechanisms responsible for various processes occurring in the brain, we uncovered a novel role for the axon guidance molecule Robo2 in excitatory synapse formation in the mouse hippocampus. Cell-autonomous deletion of Robo2 from CA1 pyramidal neurons (PNs) leads to a drastic reduction of the number of excitatory synapses specifically in proximal dendritic compartments. We show that this postsynaptic function of Robo2 depends on both its canonical ligand Slit and a novel interaction with presynaptic Neurexins.
To explore the distribution of CA1 pyramidal neurons in mice during spatial navigation, 2-photon (2P) and calcium (Ca2+) imaging was used. We found that a sparse deletion of Robo2 during development drastically alters the spatial coding properties of CA1 PNs.
Our results identify Robo2 as a novel effector linking synaptic specificity to circuit function.
Find out more about the technology featured in this webinar or our other solutions for Multiphoton Microscopy:
Featured Products
Speakers
Dr. Heike Blockus
K99-NINDS fellow at Columbia University
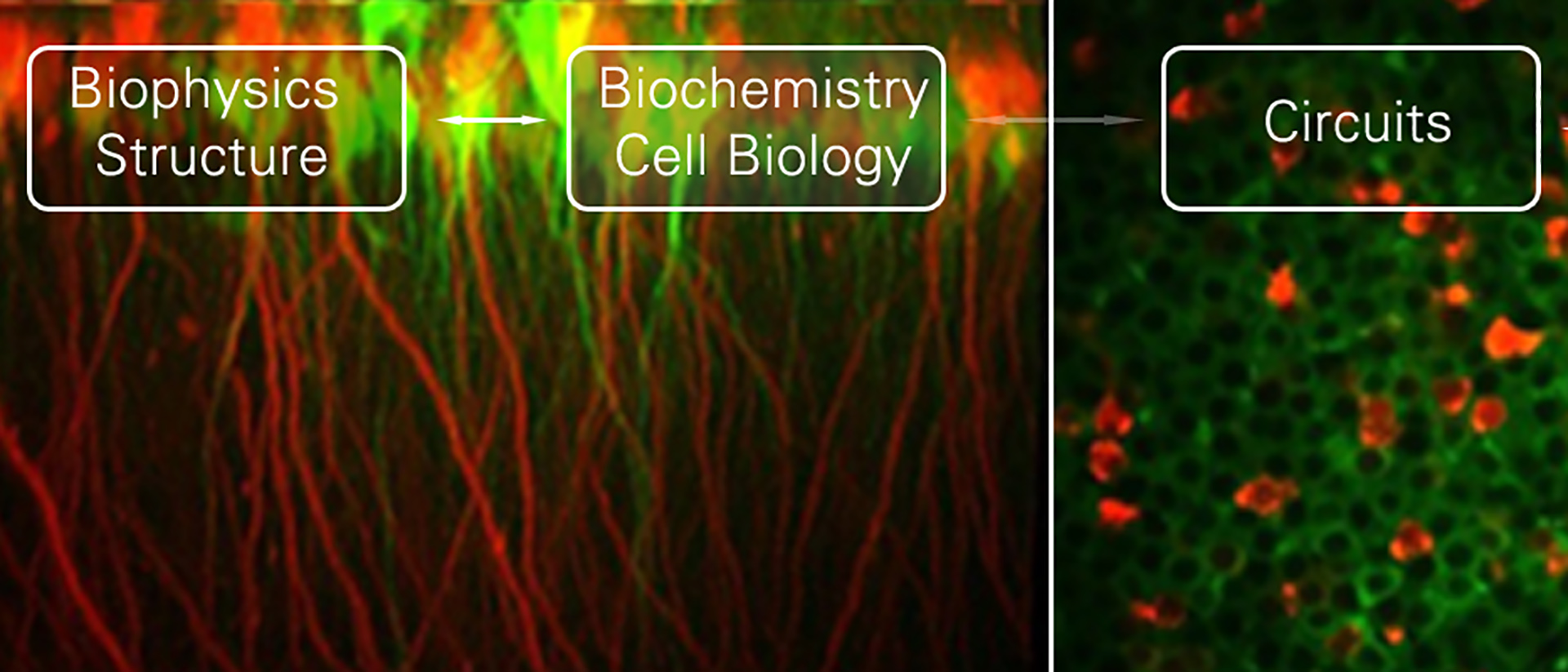
Dr. Blockus studied Molecular Biomedicine at the University of Bonn, Germany, before completing her PhD at the Sorbonne University in Paris, France, with Dr. Alain Chedotal. As a Postdoctoral Researcher and recent K99 awardee, she is co-mentored by Drs Franck Polleux and Attila Losonczy at Columbia University in New York. Dr. Blockus is interested in understanding how molecular effectors of synaptic specificity contribute to neuronal circuit function. She uses the rodent hippocampus to study how developmental molecules determine synaptic connectivity and how this relates to spatial memory formation and stability. Her approach covers a wide range of techniques from biophysics and structural biology over cell biology and anatomy to in vivo two-photon calcium imaging in awake-behaving mice.