
AST / MIC / Resistance Testing
AST / MIC / Resistance Testing
Detecting the potential resistance mechanisms and phenotypic resistance of organisms with high mortality rates such as Enterococcus faecium, Staphylococcus aureus, Klebsiella pneumoniae, Acinetobacter baumannii, Pseudomonas aeruginosa, Enterobacter spp. and Escherichia coli is of high clinical and epidemiological importance.
Bruker offers a meaningful portfolio of MALDI-TOF based, broth microdilution and molecular assays to detect antimicrobial resistance determinants and phenotypic resistance to critically important antibiotics.
MALDI-TOF mass spectrometry has the potential to identify specific resistance markers. When using the IVD MALDI Biotyper® the dedicated MBT Subtyping® IVD Module combines the identification of important pathogens with automated subsequent detection of specific resistance markers in one automated workflow, providing a warning about a potential resistance.
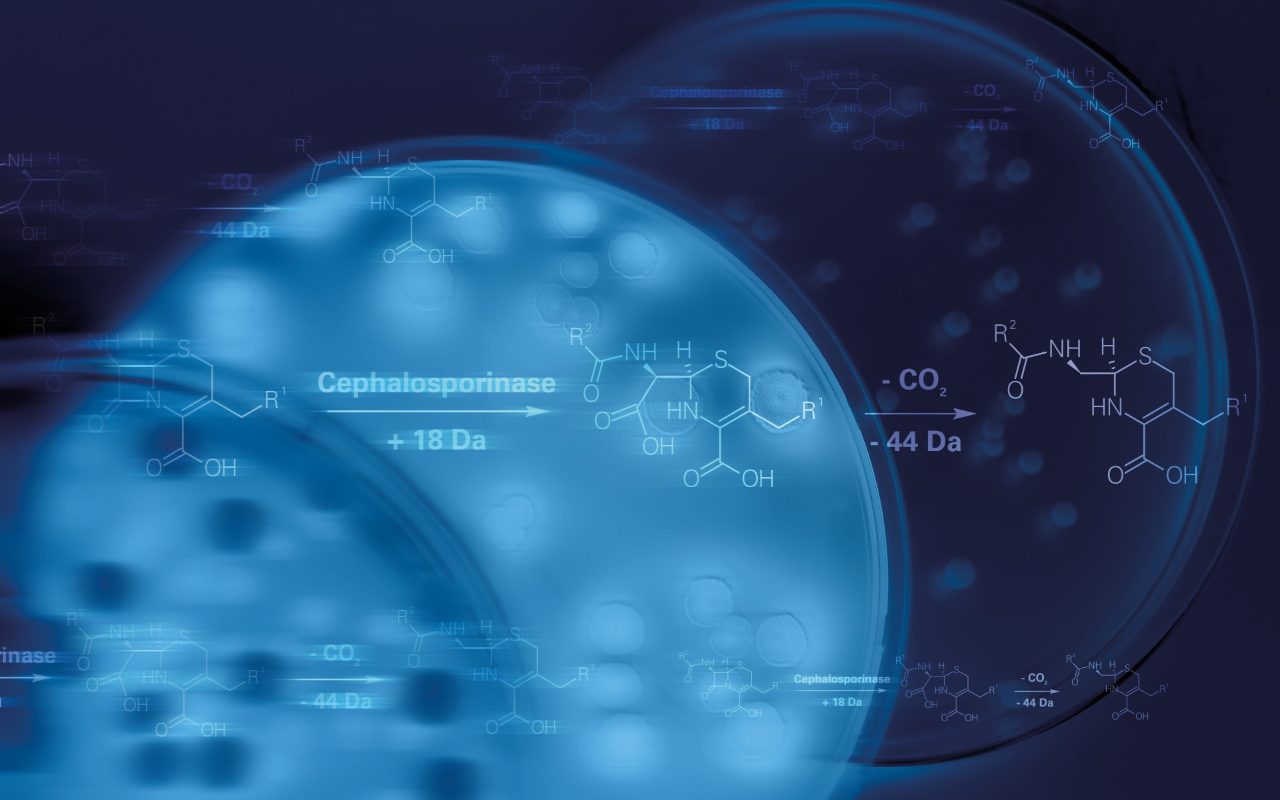
With the subsequent MBT-STAR® IVD assays, the IVD MALDI Biotyper® combines microbial identification with phenotypic detection of carbapenemase and cephalosporinase activity.
With the negative ion mode available on Bruker’s high-end MALDI Biotyper® sirius Systems, analysis of lipids has become accessible for microbial research, enabling exploration of lipidomics for resistance detection. The MBT Lipid Xtract™ Kit (RUO) allows sample preparation of the Lipid A molecule and all its modifications within less than 15 minutes and therefore delivering rapid colistin resistance detection.
The MICRONAUT and UMIC® broth microdilution (BMD) assays provide specialty and routine antimicrobial and antifungal susceptibility testing solutions as well as convenient and flexible MIC determination of highest accuracy for single antibiotics (UMIC®) like colistin, cefiderocol, and piperacillin-tazobactam.
Discover exclusive insights with our newsletter!
Get behind-the-scenes stories, explore the latest diagnostic technologies, and benefit from expert insights. Stay informed and ahead - subscribe now and join our community!