DIFFRAC.SUITE Software Family
The DIFFRAC.SUITE software guides novice users through streamlined method development and execution while assisting experts in an unobtrusive manner. The fully customizable user-interface is characterized by an ergonomic plug-in framework, providing for common look & feel and operation of all current generation XRD instruments.
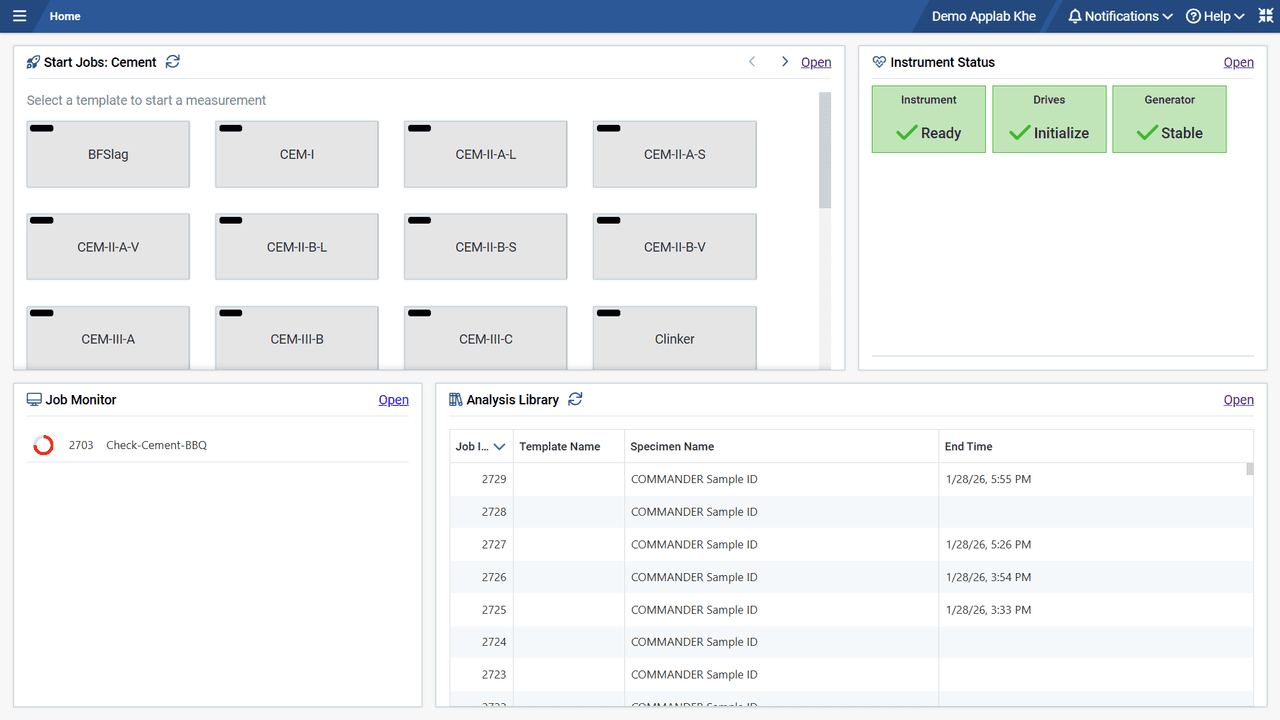
From the MEASUREMENT CENTER our D6 PHASER, D8 ENDEAVOR, and D8 ADVANCE and DISCOVER family instruments can be operated fully interactive by the expert while at the same time a simple push-button and touch-control operation makes them accessible to any operator.
The same idea holds for the data analysis part of DIFFRAC.SUITE. It covers anything between automated data evaluation after a measurement is finished, to fully interactive evaluations by the X-ray specialist. Available packages comprise DIFFRAC.EVA for general data evaluation, powder analysis software (TOPAS, DQUANT, SAXS), materials research analysis Software (LEPTOS, TEXTURE), and the thin-film analysis software LEPTOS X.
Highlights
Operation Modes
The DIFFRAC.SUITE family is based on a client/server architecture and supports unlimited networking. The software
- allows access and control of any number of D6 PHASER, D8 ADVANCE, D8 DISCOVER and D8 ENDEAVOR diffractometers within a customer's network,
- provides the opportunity for BRUKER service to remote diagnose the hardware,
- enables our application experts to maintain customer specific applications.
The “island mode” provides stand-alone operation without the need for a network. The platform concept of DIFFRAC.SUITE also allows for data exchange with our elemental analyzers.
For the Compliant Lab – 21CFR Part 11
Our software is being developed by applying a formal design process and product development life cycle according to Bruker AXS's ISO9001 certified product development procedures. Written standards exist such as coding standards, configuration management, programmer qualifications, software version control, maintenance, formal testing of software/hardware, incident reporting and tracking, and disaster recovery.
To integrate into an FDA’s 21CFR Part 11 (or other) compliant laboratory, DIFFRAC.SUITE Part 11 software offers tools to provide and guarantee authenticity, integrity and confidentiality of electronic records and electronic signatures including: secure system log-ins, automatic audit trail generation, electronic signatures with reports and data, network security, and tamper proof data files with the ability to discern invalid or altered records.
DIFFRAC.SUITE Specifications
Platform | Microsoft .NET | |
Operating system | Windows 10 and 11, 64 bit | |
Data storage | Filesystem and SQL database 21 CFR Part 11 database mode only |