Advanced Chemical Profiling 2.0
Highlights
End‑to‑End Workflow
Advanced Chemical Profiling 2.0: One software platform for quantitative NMR Workflows at any field strength
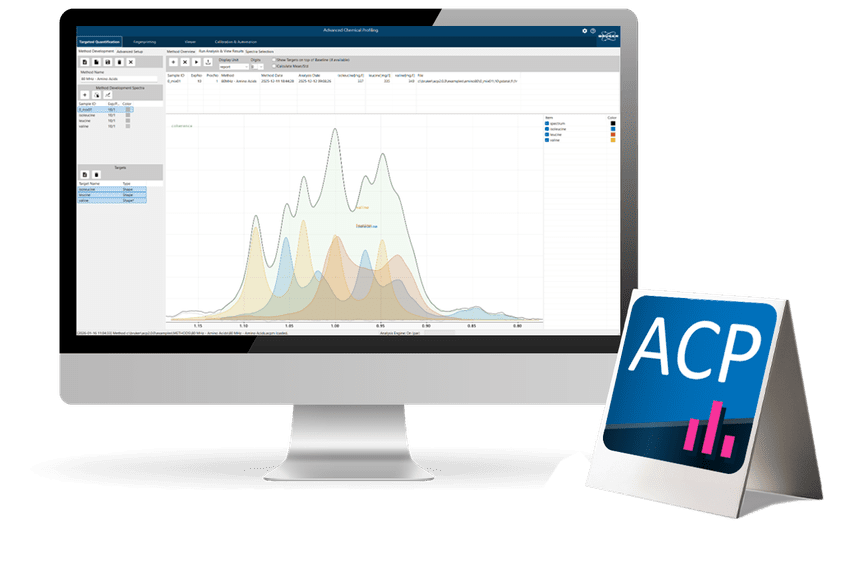
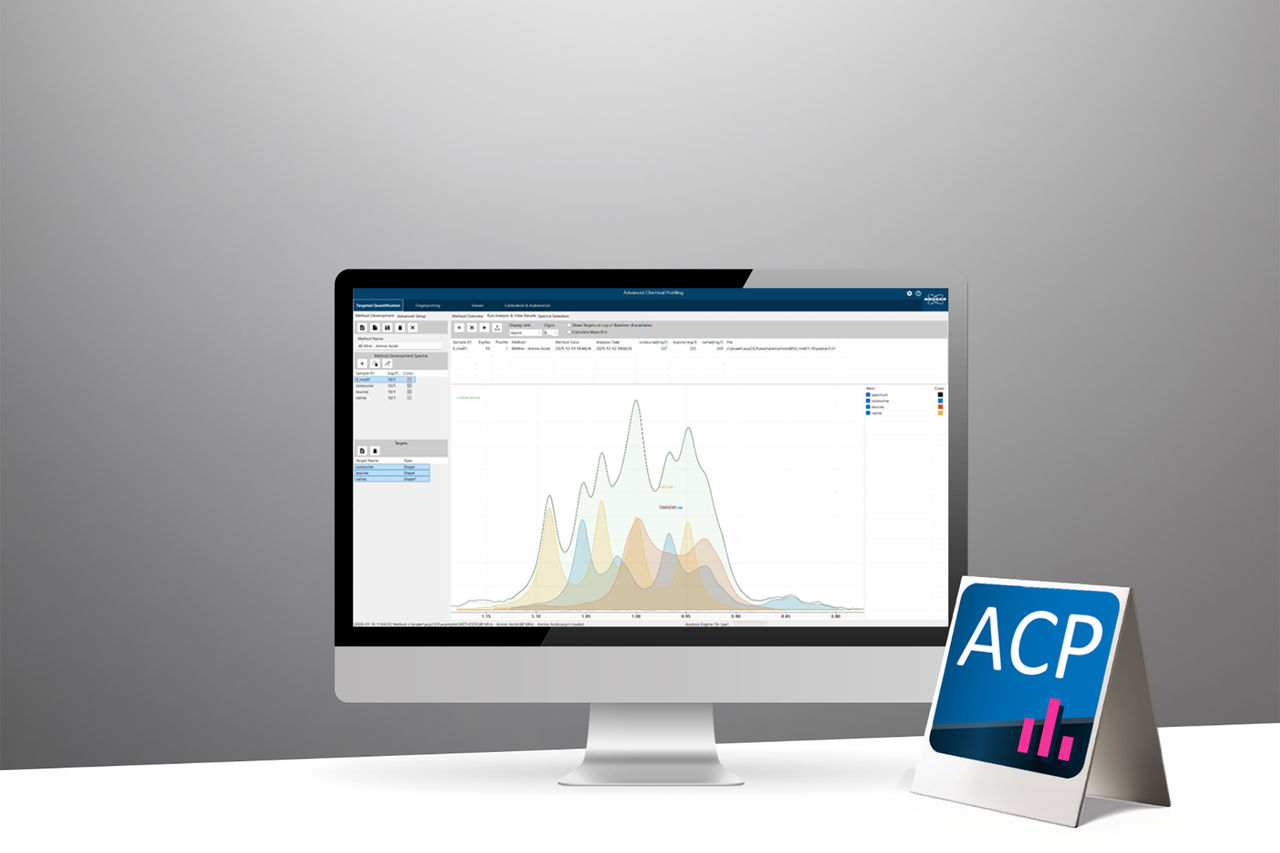
Advanced Chemical Profiling 2.0 is Bruker’s next‑generation software designed to transform quantitative NMR analysis into a fully automated, scalable, and operator‑independent workflow.
It helps laboratories harmonize analytical methods across research, development, manufacturing, and quality control. The fully automated, end‑to‑end NMR workflow increases throughput and efficiency while reducing costs through standardized, GMP‑ready, operator‑independent analysis
ACP 2.0 delivers consistent, high‑quality results by eliminating manual interpretation, enabling confident use in both expert and non‑expert environments. Its standardized pass/fail logic, shareable methods, and seamless integration with Bruker NMR systems and TopSpin™ ensure reproducible outcomes across instruments, sites, and global operations.
With intuitive setup, advanced algorithms, and broad application coverage, ACP 2.0 unlocks deep analytical insights while simplifying everyday work, supporting labs across chemical industry, food, pharmaceutical quality control and development workflows, and academic metabolomics research.
Enabling concepts like Lab-of-the-Future, it unites automation, harmonization, and usability to drive scientific excellence and operational efficiency at scale.
Software Download
Advanced Chemical Profiling 2.0
- Download software and comprehensive tutorial videos
- Install without administrator privileges
- Familiarize yourself with the capabilities of ACP 2.0 using the out-of-the-box example datasets
- Contact us for a free-of-charge 90-day demo license (Request demo license)
Discover Tutorials
You will find step-by-step video tutorials in the installation folder about:
- Installation and Testing
- Installation and Testing for GxP Readiness
- Calibration Setup
- Targeted Quantification
- Fingerprinting
- Extended Configuration
- Report Customization
- Viewer for Spectra Inspection
Features
Workflow Automation – at any Field
- Fully Automated Workflow
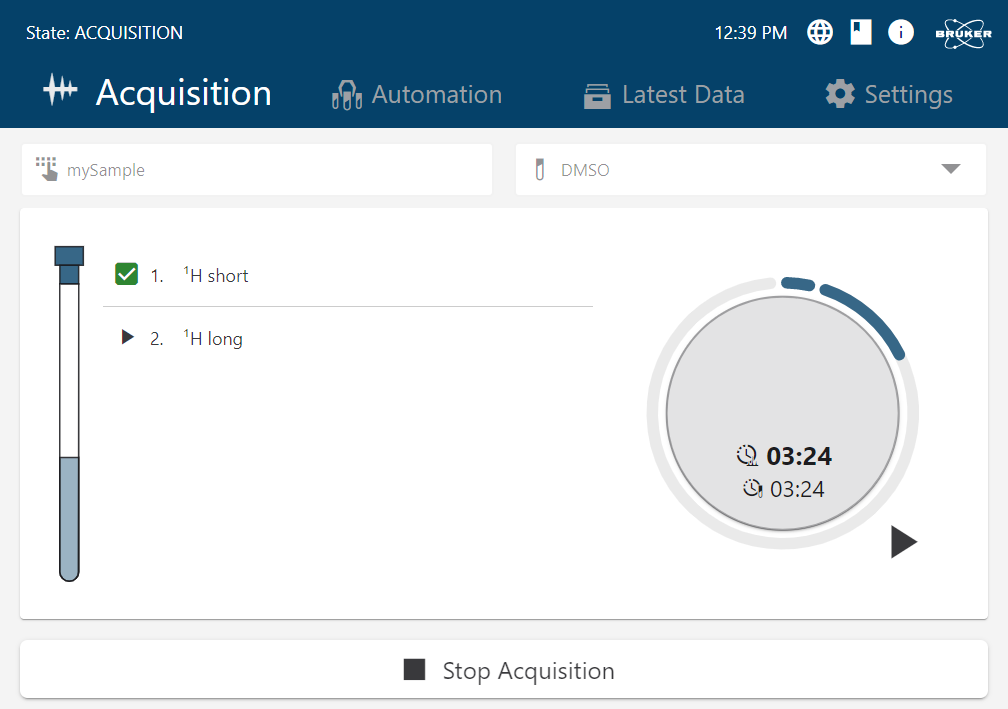
ACP 2.0 delivers a seamless, end-to-end workflow - from sample submission to final report - eliminating manual steps and ensuring consistent, operator-independent results across any skill level. - One Software Platform. All Bruker NMR
Compatible with both benchtop and high-field Bruker NMR systems, the software ensures high-performance analyses at any field and effortless rollout across R&D, QC, and manufacturing sites. - Integrated with TopSpin, lconNMR, SpinPilot & GoScan
ACP 2.0 seamlessly merges acquisition, processing, data analysis, and reporting, enabling hands-off operation without additional training. Operators interact only with familiar Bruker software environments. - Powerful Batch & On-the-Fly Processing
Analyze multiple samples or time-course datasets in one run. Batch tools increase throughput, while on-the-fly analysis provides immediate insights after measurement.
Reporting made easy – customized, standardized, compliant
- Instant, Compliant Reporting
Generate clear, detailed PDF reports or export structured machine-readable files to feed LIMS systems, empowering real‑time data use and supporting regulatory GMP/GLP compliance. - Customizable Reporting & Calculations
Adjust report formats and apply custom calculation rules (e.g., purity, monomer ratios, total sugar), eliminating the need for external tools and unifying all outputs in one system. - Standardized Pass/Fail Decisions
Embedded decision logic automates assessments for incoming goods, in-process control, and batch-release - accelerating decision-making while ensuring full harmonization across sites.
From simple to complex – for 1H and any X-nuclei
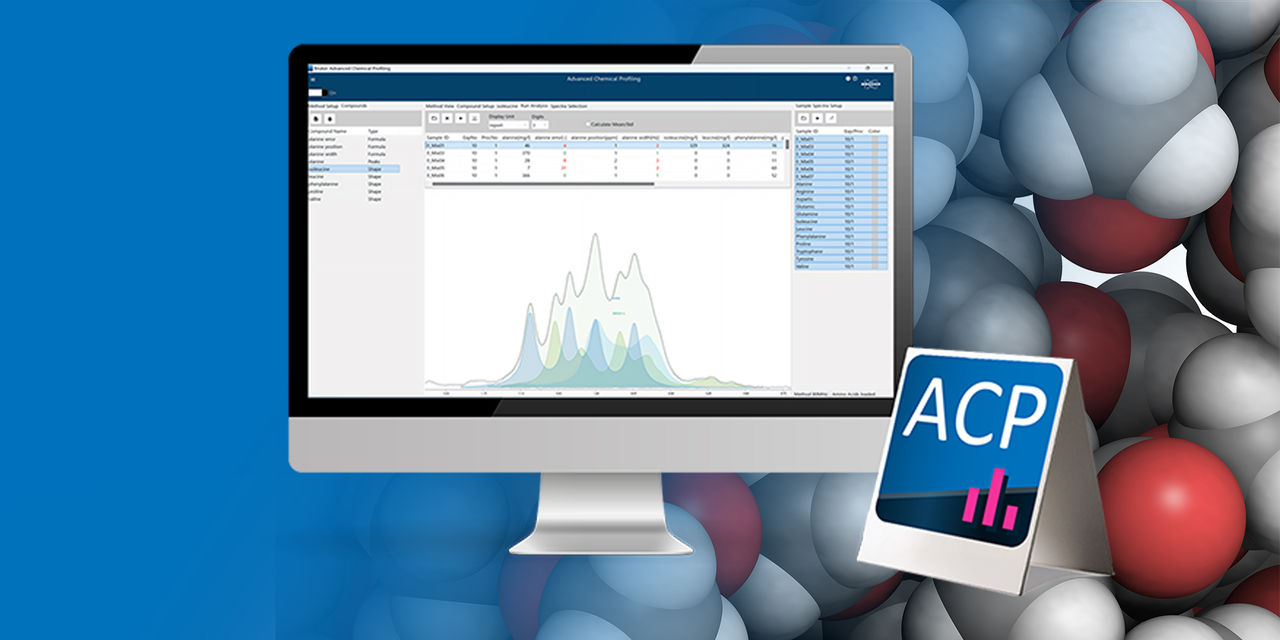
- Automated Identification & Quantification
Advanced algorithms perform identification and quantification without expert interpretation, enabling novices to execute high-value analyses and ensuring cross-site comparability. - Powerful Tools for Complex Spectra
Advanced region integration, smart modelling, signal fitting and baseline handling enable precise analysis even in challenging samples with overlaps or macromolecular background. - X·Nuclei Ready
Automate identification and quantification for X-nuclei 1 D-NMR spectra, unlocking valuable information beyond standard proton NMR.
Harmonize – globally
- Shareable Methods & Databases
Methods, compounds and databases defined in ACP 2.0 can be shared across teams, authorities, or sites, enabling harmonized deployment and reducing duplication of work.
Benefits
- Automation & Accessibility
ACP 2.0 delivers fully automated, end-to-end NMR workflows that eliminate manual intervention, enabling consistent, high-quality analysis even in non-expert environments. - High Throughput & Efficiency
ACP 2.0 accelerates analytical workflows through on-the-fly analysis or batch processing as well as streamlined data handling, significantly increasing throughput and freeing up expert time. - Standardization & Decision Support
ACP 2.0 empowers standardized, data-driven decision-making by automating pass/fail assessments for critical control points in chemical production. - Knowledge Sharing & Harmonization
ACP 2.0 facilitates seamless sharing of analytical methods across teams and sites, driving harmonization and collaboration in global organizations. - Usability & User Experience
ACP 2.0 combines powerful analytical capabilities with an intuitive interface, enabling users to set up sophisticated NMR workflows with just a few clicks.
Unlocking NMR Potential: Elevating NMR Beyond Structural Elucidation
Research & Development
ACP 2.0 provides deep chemical insight for R&D teams by automating identification, quantification, and characterization workflows. It supports rapid formulation work, impurity profiling, stability studies, and method development by extracting rich information from even complex spectra. Scientists gain faster, clearer answers and can focus their time on innovation rather than routine analysis.
Process & In‑Process Control
In manufacturing environments, ACP 2.0 standardizes analytical evaluation of in‑process samples, enabling rapid, data‑driven decisions during production. Automated pass/fail logic ensures consistent evaluation of critical parameters, reduces interpretation errors, and accelerates reaction monitoring, process adjustments, and troubleshooting during scale‑up or continuous production.
Quality Control & Batch Release
For QC laboratories, ACP 2.0 guarantees harmonized, operator‑independent results across global sites. Automated quantification, structured reporting, and machine-readable data transfer strengthen compliance and deliver high confidence in purity checks, concentration assessments, and conformity evaluations of incoming goods, intermediates, and final products. This ensures reliable, repeatable decisions and shortens release timelines.
On‑Site and Decentralized Analysis
ACP 2.0 enables high‑performance analytical capabilities to move closer to the production floor. Compatible with both benchtop and high‑field NMR systems, it allows non‑experts to execute standardized methods on‑site, eliminating delays and improving uptime. Plants can benefit from immediate results, reduced back‑and‑forth with central labs, and more resilient operations.
Beyond Automation: Analytical Tool to Analyze Even Most Complex Mixture
With smart modelling, coherent fitting, baseline handling, and X‑nuclei support, ACP 2.0 readily tackles challenging samples such as polymers, fermentation media, specialty chemicals, and bioprocessing fluids – samples that are difficult to analyze manually. This unlocks insights that often require multiple orthogonal methods - enabling deeper understanding and faster troubleshooting.
Use Case Examples
Specifications
Discover some features examples in the carousel
ACP 2.0 Launch Discount
Limited‑time launch offer ready for you : 50% off ACP 2.0 for licenses purchased before August 31st, 2026*.
*Available for annual licenses only.
Testimonials
Case study - WACKER is driving research in the field
of industrial carbon capture and utilizationAdvanced Chemical Profiling Module helps to generate robust, reproducible quantifications of the measured data. As soon as a database is set up accordingly, the process of using the new software module does not require NMR expert knowledge. This procedure should be well suited for production environments where the contents of the samples are comparable and the sample matrices are not too complex.
dsm-firmenich Science & Research
At dsm-firmenich, we’re at the forefront of innovation in nutrition, health, and beauty. We create sustainable solutions that impact our daily lives. Our cutting-edge approaches drive progress, with one area of focus being the development of novel bioprocesses.
In our R&D lab, we regularly use Nuclear Magnetic Resonance (NMR) to screen new fermentation processes. These advanced bioprocesses involve complex mixtures of media components and metabolites. Our goal is to gain an in-depth understanding and enable data-based development of new procedures.
Through our long-standing partnership with Bruker, we have been testing the latest software release from their Advanced Chemical Profiling Solution. This supports the development of the kinds of analytical workflows we use, seamlessly integrating data acquisition, processing, interpretation, and report filing. It streamlines analytical workflows and makes it easier to compare results.
Bruker’s software platforms can be used to support scale-up from development to manufacturing. As new production routes are rolled out into manufacturing, process monitoring with a benchtop NMR system would be feasible. Bruker’s Fourier 80 benchtop system, equipped with the same Advanced Chemical Profiling Solution, could support process control in production labs.
An integrated NMR methodology - combining high-field NMR in R&D labs with benchtop systems in manufacturing - opens exciting opportunities for the industry. End-to-end use of Bruker’s solutions can make it easier to develop efficient, scalable processes.
Dr. Henrik Scholz, Advanced Scientist Process Technology, Momentive Performance Materials GmbH
As a premier global high-performance silicones and specialties company, Momentive creates solutions that improve quality of life for people and enable a more sustainable future.
This encompasses the development of new products and processes including cutting-edge analytical methodologies to gain in-depth understanding driving data-based decision making. Nuclear Magnetic Resonance (NMR) is a powerful tool to access structural and quantitative information of some of our highly complex process samples and products. Along this line, we utilize Bruker’s next-generation quantitative NMR workflow automation software “Advanced Chemical Profiling 2.0”, which features modelling routines that can decipher highly complex multi-component mixtures as often encountered in industrial in-process or product quality control tasks. These capabilities help us in gaining new insights into products and processes that are otherwise inaccessible or very cumbersome to obtain with alternative analytical methods. Advanced Chemical Profiling 2.0 allows to intuitively setup analytical methods for NMR data interpretation and apply the methods to dozens of spectra simultaneously which not only significantly reduces the workload for manual data interpretation but ensures to achieve operator-independent results. Importantly, key quality criteria can be assigned with pass or fail conditions implemented in the method setup which allows the results to be interpretable even by NMR novices. Moving forward, we consider embedding the developed and validated methods into fully automated workflows to achieve push-button operation from sample to report.
James Sklut, University of Tübingen, PhD student- Core Facility Metabolomics, M3 Research Center, University Hospital Tübingen
For me, the real strength of ACP 2.0 is the confidence it gives in the results. The AI macromolecular filter works extremely well on complex blood and tissue H-NOESY spectra, and when it’s combined with automated fitting, the annotations are simply much more reliable. What used to take the time and effort of a full project can now be done across multiple projects, and with the flexibility to include metabolites we weren’t originally screening for. That changes how much data we can realistically analyze - and trust - within a given timeframe.
Dr. Matthias Abele, Head of EHSQ & Sustainability, Evonik
Bruker’s new Advanced Chemical Profiling software can significantly enhance the mixture analysis process due to its robust and accurate identification and quantification feature. The complete integration into the instrument control software TopSpin and the sample automation software IconNMR™ allows for comprehensive workflow creation from the sample in the queue till the analysis report. Various concentration units and intuitive internal and external reference management makes the new software suitable for many existing analytical workflows. Once the individual methods are validated and implemented into the automated workflow, the push-button operation and operator-independent data interpretation matches industrial product quality control requirements.
Distributed Lab Topology Environment
Globally organized companies might request these duties from their labs
- Method developed on R&D High-Resolution (HiRes) NMR systems
- Pilot manufacturing support by R&D department
- Volume manufacturing method development for non-expert operation (QC)
- QC method validation via R&D method
The Advanced Chemical Profiling Solution will support you in:
- Direct HiRes NMR method roll-out to QC if needed
- HiRes NMR to benchtop NMR method translation if possible
- Direct (HiRes/HiRes) or In-Tech (HiRes/Benchtop) method validation decreases time-to-market
- Database-driven method scaling opportunity to other product lines (same instrument possible)
Download Distributed Lab Topology eBook:
LabScape
Service & Life Cycle Support for Magnetic Resonance and Preclinical Imaging
Bruker’s commitment to provide customers with unparalleled help throughout the buying cycle, from initial inquiry to evaluation, installation, and the lifetime of the instrument is now characterized by the LabScape service concept.
LabScape Maintenance Agreements, On-Site On-Demand and Enhance Your Lab are designed to offer a new approach to maintenance and service for the modern laboratory