Preclinical Imaging
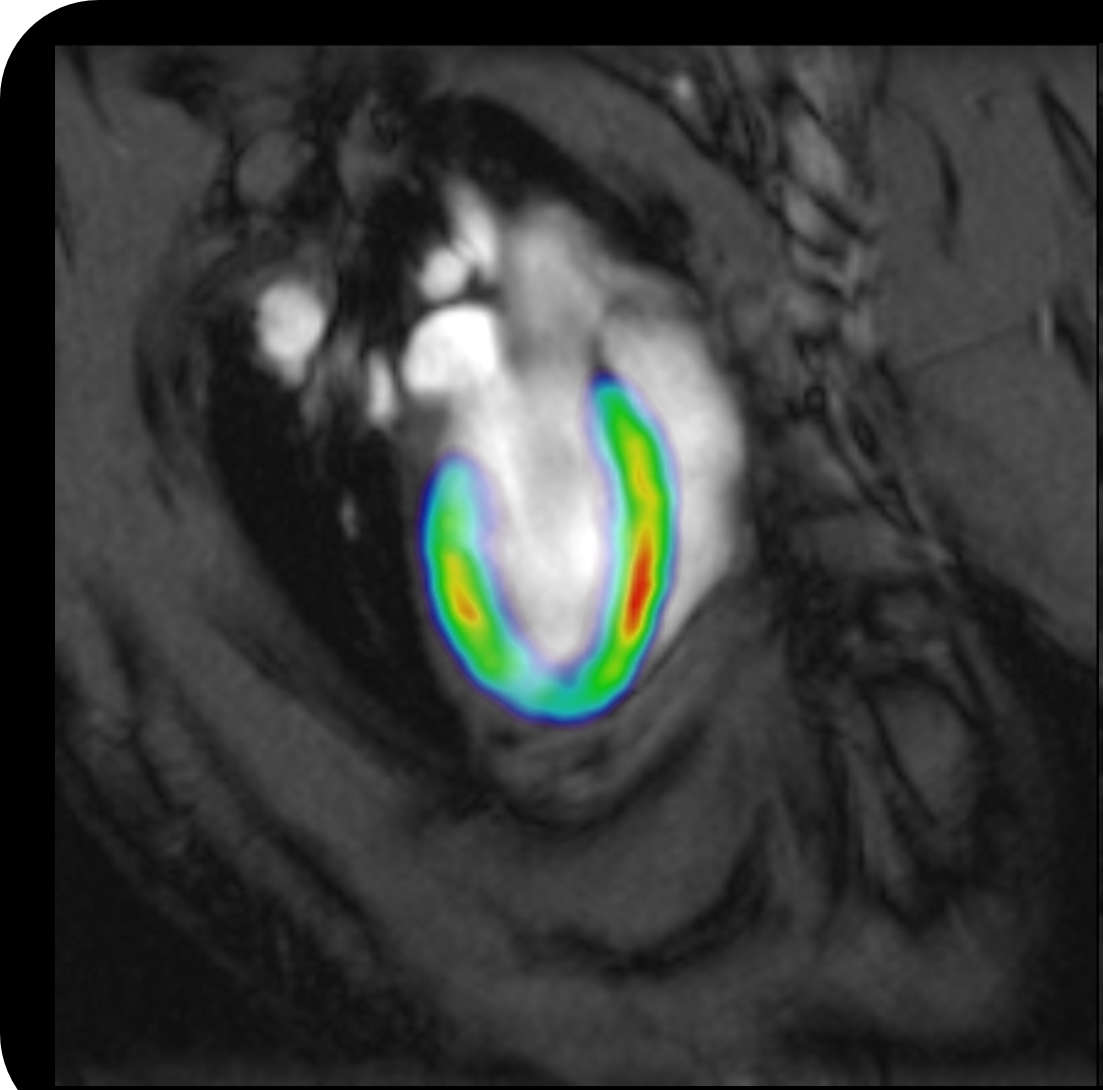
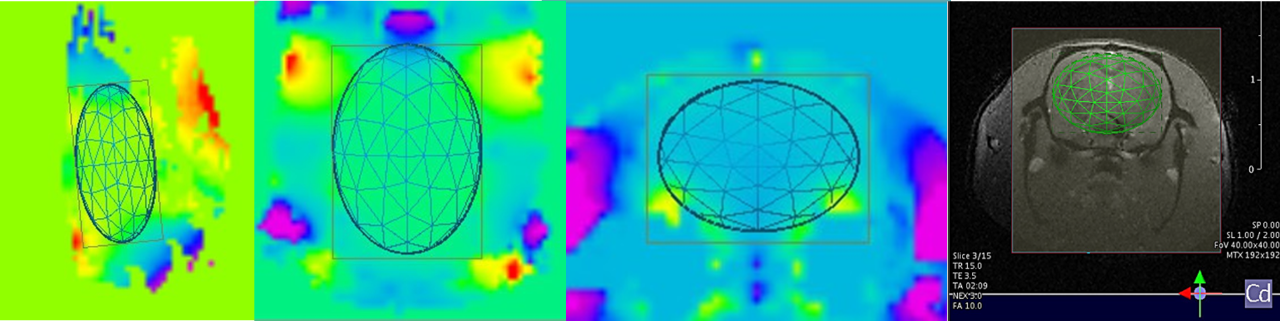
Bruker offers advanced preclinical imaging solutions for a broad spectrum of application fields, such as oncology, neurology, cardiology, inflammation, infectious diseases, cancer research, functional and anatomical neuroimaging, orthopedics, cardiac imaging and stroke models.
Preclinical Imaging Instruments
Multimodal Solutions Empowering Breakthroughs
in Life Science Research
LabScape
Service & Life Cycle Support for Magnetic Resonance and Preclinical Imaging
Bruker’s commitment to provide customers with unparalleled help throughout the buying cycle, from initial inquiry to evaluation, installation, and the lifetime of the instrument is now characterized by the LabScape service concept.
LabScape Maintenance Agreements, On-Site On-Demand and Enhance Your Lab are designed to offer a new approach to maintenance and service for the modern laboratory